http://www.muscle_____chat_____room.com/forum/showthread.php?t=18
null good starting page
http://www.meta-ehealth.com/site/office/print.jsp?path=fm_wellness_center/nutrition/nutritional_health_encyclopedia&id=6
Nutritional Influences on Estrogen Metabolism: A Summary
Nutritional Influences on Estrogen Metabolism: A Summary
By Douglas C. Hall, M.D.
Estrogen affects the growth, differentiation, and function of tissues throughout the body?not just those involved in reproduction. It plays an important role in bone health, protects the cardiovascular system, and influences behavior and mood. While appropriate levels of estrogens are essential for good health, several studies conclude that as exposure to estrogen increases, the risk of several cancers, including breast, ovary, prostate, and thyroid, also increases.1-6 Furthermore, excessive estrogen exposure can lead to other health problems such as premenstrual syndrome (PMS), endometriosis, and fibrocystic or painful breasts.
Various lifestyle and environmental factors can influence estrogen production, metabolism, and balance. These include poor diet, obesity,excess alcohol consumption, high insulin levels, medications such as hormone replacement therapy and birth control pills, overexposure to chemicals found in pesticides and industrial chemicals, and agricultural hormones in animal products consumed by humans.2,7-11 Genetics can also play an important role in determining estrogen levels.
The Basics of Estrogen Metabolism
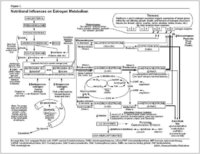
"Estrogen" is a term that is used to collectively describe the female hormones estradiol, estrone, and estriol. The most potent of these is estradiol. Estrogens circulate in the body mainly bound to the sex hormone binding globulin (SHBG) and only unbound estrogens can enter cells and cause biological effects.12,13 Therefore, any change in the concentration of SHBG will alter estrogen activity by changing the availability of estrogen to the target cell.
The ultimate biologic effect of estrogen in the body depends on how it is metabolized. The metabolism of estrogen takes place primarily in the liver through Phase I (hydroxylation) and Phase II (methylation and glucuronidation) pathways, which allow the estrogen to be detoxified and excreted from the body.
Hydroxylation ?Hydroxylation yields three metabolites that vary greatly in biological activity: 2-hydroxyestrone (2-OH),16-OH, or 4-OH.14 The 2-OH metabolite is generally termed the "good" estrogen because it generates very weak (and therefore potentially less harmful) estrogenic activity in the body. In contrast, the 16-OH and 4-OH metabolites show persistent estrogenic activity and may promote dangerous tissue growth.14-17 In fact, women who metabolize a larger proportion of their estrogen via the 16-OH metabolite may be at significantly greater risk of developing breast cancer.1,14-16,18,19 Therefore, shifting estrogen balance toward a less estrogenic state through promotion of the 2-OH pathway may prove very beneficial in improving a variety of conditions related to elevated or imbalanced estrogen levels.
Methylation ?The 2-OH and 4-OH estrogen metabolites are further detoxified via a process called methylation. This is an important pathway, because it renders the harmful 4-OH metabolite significantly less active. Furthermore, if they are not methylated, the 2-OH and 4-OH estrogens can be converted to highly reactive molecules that can damage DNA.16,20,21
Glucoronidation ?In glucoronidation, a glucuronic acid group combines with an estrogen molecule to facilitate the elimination of excess estrogen from the body.12 These actions make glucoronidation one of the key Phase II liver detoxification pathways for estrogens.
Nutritional Support of Optimum Estrogen Metabolism
Many elements of good nutrition and diet play an important part in influencing estrogen metabolism and detoxification. Incorporating dietary changes with the addition of beneficial nutrients and herbs can profoundly affect estrogen balance and potentially reduce the risk of estrogen-dependent cancers and other hormone-related conditions.
Diet?It has been found that dietary interventions such as increasing consumption of cruciferous vegetables like cabbage and broccoli, and foods such as soy can significantly increase the 2-hydroxylation of estrogen. Dietary fiber intake can promote the excretion of estrogen by binding estrogens in the digestive tract and also increases serum concentrations of SHBG, thus reducing levels of free estradiol.22,23 Complex carbohydrates, such as those found in vegetables and whole grains, are more effective in optimizing estrogen metabolism than simple carbohydrates, which can detrimentally raise blood glucose levels and stimulate insulin release, resulting in secondary adverse influences on sex hormone balance.8
Phytoestrogens?These plant compounds are similar in shape to the estrogen molecule and can bind to estrogen receptors (ERs). They are much weaker than endogenous estrogens and, through competitive inhibition, have been shown to prevent the receptor binding of "stronger," more stimulating estrogens.7,24,25 Phytoestrogens are currently under extensive investigation as a potential alternative therapy for a range of conditions associated with estrogen imbalance, including menopausal symptoms, PMS, endometriosis, prevention of breast and prostate cancer, and protection against heart disease and osteoporosis.7,25-27
The two main classes of phytoestrogens are isoflavones and lignans. Soy is perhaps the most common food source of isoflavones, but other excellent sources include legumes, clover, and kudzu root. Higher intakes of soy products and isoflavones, such as consumed in traditional Japanese diets, are associated with low rates of hormone-dependent cancers.28 Lignans are compounds are found in fiber-rich foods such as flaxseed and other oil seeds, whole grains, legumes, and vegetables.29,30 Lignans stimulate the production of SHBG in the liver, and therefore reduce the levels of free estrogen in circulation. They also inhibit aromatase, an enzyme that synthesizes estrogen.
Vitamin E and Magnesium?Low serum vitamin E is associated with elevated estrogen levels, and may negatively affect estrogen detoxification. Women with PMS have experienced improvements of their symptoms when given supplemental vitamin E.31 Magnesium promotes estrogen detoxification by promoting methylation and glucuronidation, key estrogen detoxification pathways. Ovarian hormones influence magnesium levels, triggering decreases at certain times during the menstrual cycle as well as altering the calcium to magnesium ratio. These cyclical changes can produce many of the well-known symptoms of PMS in women who are deficient in magnesium and/or calcium.32
Indole-3-Carbinol (I3C)?I3C is a naturally occurring compound derived from cruciferous vegetables such as broccoli, Brussels sprouts, and cabbage that actively promotes the breakdown of estrogen via the beneficial 2-OH pathway.14,33-35 Therefore, I3C is protective to estrogen-sensitive tissues and may be beneficial to those with health issues related to excessive estrogen. Not only does I3C promote healthier estrogen metabolism, but it may also act as a "weak" or anti-estrogen in a similar fashion to isoflavones.36
B Vitamins?Folate and vitamins B6 and B12 function as important cofactors for enzymes involved in estrogen detoxification; thus, decreased levels of B vitamins can lead to increased levels of circulating estrogens. Certain B vitamins also have the potential to modulate the biological effects of estrogen by decreasing the cell's response when estrogen binds to the ER.37 B vitamins also play a role in the prevention of cancer because they are important for DNA synthesis and repair.
Calcium D-Glucarate?Calcium D-glucarate is a natural compound found in foods that appears to have some influence on breast cancer by aiding in detoxification and the regulation of estrogen.38,39 It has been found in animal models to lower estradiol levels and inhibit the initiation, promotion, and progression of cancer.38
Other Beneficial Phytonutrients and Herbs
Many other naturally occurring compounds derived from a variety of plant sources are available that promote healthy estrogen metabolism. These include curcumin, a compound found in the herb turmeric (Curcuma longa) that increases the phase II detoxification of catechol estrogens;40,41 chrysin, a bioflavonoid that inhibits aromatase activity, thus reducing the synthesis of estrogen activity;42 the herb, rosemary, which promotes the formation of the 2-OH estrogen metabolite;43 and D-limonene from citrus fruits, which promotes the detoxification of estrogen and shows promise in the prevention and treatment of breast and other cancers.44,45 Furthermore, many antioxidant nutrients and phytonutrients can reduce the oxidation of the 2-OH and 4-OH estrogen metabolites. Notable nutrients in this group include vitamin C, N-acetylcysteine, the mineral selenium, and green tea.
In addition, traditional societies have long relied on a variety of hormone-modulating herbs in treating women's health conditions. These include black cohosh, chasteberry, ginseng, dong quai, and licorice. The mechanism of action of these herbs varies; however, many have been found to contain beneficial phytoestrogens.
References
Bolton JL, Pisha E, Zhang F, et al. Role of quinoids in estrogen carcinogenesis. Chem Res Toxicol 1998;11:1113-27.
Colditz GA. Relationship between estrogen levels, use of hormone replacement therapy, and breast cancer. J Natl Cancer Inst 1998;90(11):814-23.
Thomas HV, Reeves GK, Key TJ. Endogenous estrogen and postmenopausal breast cancer: a quantitative review. Cancer Causes Control 1997;8(6):922-28.
Rose PG. Endometrial carcinoma. New Eng J Med 1996;335(9):640-49.
Hankinson SE, Willett WC, Manson JE, et al. Plasma sex steroid hormone levels and risk of breast cancer in postmenopausal women. J Natl Cancer Inst 1998;90(17):1292-99.
Zanetta GM, Webb MJ, Li H, et al. Hyperestrogenism: A relevant risk factor for the development of cancer from endometriosis. Gynecol Oncol 2000 Oct;79(1):18-22.
Kuiper GG, Lemmen JG, Carlsson B, et al. Interaction of estrogenic chemicals and phytoestrogens with estrogen receptor & beta. Endocrinology 1998;139(10):4252-63.
Kaaks R. Nutrition, hormones, and breast cancer: Is insulin the missing link? Cancer Causes Control 1996;7:605-25.
Snedeker SM, Diaugustine RP. Hormonal and environmental factors affecting cell proliferation and neoplasia in the mammary gland. Prog Clin Biol Res 1996;394:211-53.
Fan S, Meng Q, Gao B, et al. Alcohol stimulates estrogen receptor signaling in human breast cancer cell lines. Cancer Res 2000;60(20):5635-39.
Steingraber S. Living Downstream. Reading (MA): Addison-Wesley; 1997:248-51.
Murray RK, Granner DK, Mayes PA, et al. Harper's Biochemistry. 24th ed. Stamford (CT): Appleton & Lange; 1996.
Guyton AC. Textbook of Medical Physiology. 8th ed. Philadelphia: WB Saunders; 1991.
Bradlow HL, Telang NT, Sepkovic DW, et al. 2-Hydroxyestrone: the 'good' estrogen. J Endocrin 1996;150:S259-S65.
Muti P, Bradlow HL, Micheli A, et al. Estrogen metabolism and risk of breast cancer: a prospective study of the 2:16-hydroxyestrone ratio in premenopausal and postmenopausal women. Epidemiology 2000;11(6):635-40.
Yager JD, Liehr JG. Molecular mechanisms of estrogen carcinogenesis. Annu Rev Pharmacol Toxicol 1996;36:203-32.
Westerlind KC, Gibson KJ, Malone P, et al. Differential effects of estrogen metabolites on bone and reproductive tissues of ovarectomized rats. J Bone Miner Res 1998;13(6):1023-31.
Meilahn EN, De Stavola B, Allen DS, et al. Do urinary oestrogen metabolites predict breast cancer? Guernsey III cohort follow-up. Br J Cancer 1998;78:1250-55.
Fishman J, Osborne MP, Telang NT. The role of estrogen in mammary carcinogenesis. Ann N Y Acad Sci 1995;768:91-100.
Zhu BT, Conney AH. Is 2-methoxyestradiol an endogenous estrogen metabolite that inhibits mammary carcinogenesis? Cancer Res 1998;58:2269-77.
Butterworth M, Lau SS, Monks TJ. 17-beta-estradiol metabolism by hamster hepatic microsomes. Implications for the catechol-O-methyl transferase-mediated detoxication of catechol estrogens. Drug Metab Dispos 1996;24(5):588-94.
Shultz TD, Howie BJ. In vitro binding of steroid hormones by natural and purified fibers. Nutr Cancer 1986;8(2):141-47.
Adlercreutz H, Hockerstedt K, Bannwart C, et al. Effect of dietary components, including lignans and phytoestrogens, on enterohepatic circulation and liver metabolites of estrogens and in sex hormone binding globulin (SHBG). J Steroid Biochem 1987;27(4-6):1135-44.
Cassidy A. Potential tissue selectivity of dietary phytoestrogens and estrogens. Curr Opin Lipidol 1999;10:47-52.
Brzezinski A, Debi A. Phytoestrogens: the "natural" selective estrogen receptor modulators? Eur J Obstet Gynecol 1999;85:47-51.
Lissin LW, Cooke JP. Phytoestrogens and cardiovascular health. J Am Coll Cardiol 2000;35(6):1403-10.
Knight DC, Eden JA. A review of the clinical effects of phytoestrogens. Obstet Gynecol 1996;87(5):897-904.
Messina MJ, Persky V, Setchell KD, et al. Soy intake and cancer risk: a review of the in vitro and in vivo data. Nutr Cancer 1994;21:113-31.
Kirkman LM, Lampe JW, Campbell DR, et al. Urinary lignan and isoflavonoid excretion in men and women consuming vegetable and soy diets. Nutr Cancer 1995;24(1):1-12.
Thompson LU, Robb P, Serraino M, et al. Mammalian lignan production from various foods. Nutr Cancer 1991;16(1):43-52.
London RS, Murphy L, Kitlowski KE, et al. Efficacy of alpha-tocopherol in the treatment of the premenstrual syndrome. J Reprod Med 1987;32:400-04.
Muneyvirci-Delale O, Nacharaju VL, Altura BM, et al. Sex steroid hormones modulate serum ionized magnesium and calcium levels throughout the menstrual cycle in women. Fertil Steril 1998;69(5):958-62.
Michnovicz JJ, Adlercreutz H, Bradlow HL. Changes in levels of urinary estrogen metabolites after oral indole-3-carbinol treatment in humans. J Natl Cancer Inst 1997;89(10):718-23.
Tiwari RK, Guo L, Bradlow HL, et al. Selective responsiveness of human breast cancer cells to indole-3-carbinol, a chemopreventive agent. J Natl Cancer Inst 1994;86(2):126-31.
Michnovicz JJ, Bradlow HL. Altered estrogen metabolism and excretion in humans following consumption of indole-3-carbinol. Nutr Cancer 1991;16(1):59-66.
Yuan F, Chen DZ, Liu K, et al. Anti-estrogenic activities of indole-3-carbinol in cervical cells: implication for prevention of cervical cancer. Anticancer Res 1999;19(3A):1673-80.
Tully DB, Allgood VE, Cidlowski JA. Modulation of steroid receptor-mediated gene expression by vitamin B6. FASEB J 1994;8(3):343-49.
Minton JP, Walaszek Z, Schooley W, et al. Beta-glucuronidase levels in patients with fibrocystic breast disease. Breast Cancer Res Treat 1986;8:217-22.
Walaszek Z, Szemraj J, Narog M, et al. Metabolism, uptake, and excretion of a D-glucaric acid salt and its potential use in cancer prevention. Cancer Detect Prev 1997;21(2):178-90.
Goud VK, Polasa K, Krishnaswamy K. Effect of turmeric on xenobiotic metabolising enzymes. Plant Foods Hum Nutr 1993;44(1):87-92.
Susan M, Rao MN. Induction of glutathione S-transferase activity by curcumin in mice. Arzneimittelforschung 1992;42(7):962-64.
Jeong HJ, Shin YG, Kim IH, et al. Inhibition of aromatase activity by flavonoids. Arch Pharm Res 1999;22(3):309-12.
Zhu BT, Loder DP, Cai MX, et al. Dietary administration of an extract from rosemary leaves enhances the liver microsomal metabolism of endogenous estrogens and decreases their uterotropic action in CD-1 mice. Carcinogenesis 1998;19(10):1821-27.
Maltzman TH, Christou M, Gould MN, et al. Effects of monoterpenoids on in vivo DMBA-DNA adduct formation and on phase I hepatic metabolizing enzymes. Carcinogenesis 1991;12:2081.
Vigushin DM, Poon GK, Boddy A, et al. Phase I and pharmacokinetic study of D-limonene in patients with advanced cancer. Cancer Chemother Pharmacol 1998;42:111-17.
Advanced Nutrition Publications ©2002
--------------------------------------------------------------------------------