
Effervescent Elitism
Advanced Muscle Science never stops bringing you the best prohormone technology. Today we are introducing you to our new Chrome Series. The Chrome Series in its simplest form utilizes the same effective rapid dissolve complex that the previous versions utilized, but introduces an effervescent matrix. Are you foaming at the mouth yet? (Pun intended)

The effervescent matrix was actually added to accomplish a few very simple things. First by adding an effervescent matrix to an orally disintegrating tab, you cause a release of carbon dioxide when it comes in contact with your saliva. This shifts the local pH as the tab disintegrates and the lower pH allows a poorly water soluble prohormone to become significantly more water soluble (dissolution). Second this enhances buccal penetration along with the use of our existing hydroxypropyl-β-cyclodextrin (HPBCD). In other words, we are getting even more prohormone directly into the bloodstream and bypassing negative hepatic metabolism. The new effervescent matrix, together with our past delivery technology, leads to improved gains, and a better bang for your buck.
To give you a better idea of how this delivery technology is superior, let’s first take a look at some current supplements and drugs using an effervescent matrix. First in 2010 research was published out of Cairo University that developed and compared an orally disintegrating tablet (including hydroxyl propyl beta cyclodextrin, and an effervescent matrix) to a regular oral supplement (1). The supplement is a popular nootropic known as vinpocetine, used to enhance memory and also sold as a drug to treat stroke victims (caviton). Vinpocetine, like prohormones is poorly water soluble and is rapidly broken down by first pass metabolism and for this reason they felt an orally disintegrating tab would improve the pharmacokinetics. And they were right. They found that the orally disintegrating tab utilizing an effervescent matrix resulted in a 306% increase in bioavailability!

Now the vinpocetine example is exciting, but it doesn’t really show us an improvement in a cyclodextrin delivery vs. a cyclodextrin + effervescent matrix. Therefore we looked to a patent filed in 2008 (2). What is great about this patent are the research and the compound they studied. The compound is known as progesterone, which is used in hormonal replacement therapy for women. Of course we don’t want to supplement progesterone, but much like prohormones progesterone is a sex steroid, which makes it poorly soluble in water. And just like prohormones progesterone has a poor bioavailability (6-8%) when administered orally (3). The inventors were trying to better an existing orally disintegrating formulation which consisted of progesterone, HPCD, as wells as other basic ingredients such as flavoring. So they created the same formula, with the addition of an effervescent matrix (citric acid, and sodium bicarbonate). With the two formulations they then completed a pharmacokinetic study using each formulation which contained equal amounts of the studied active (in this case progesterone). As you can see below there was a significantly greater spike in “Example 2” which contains the effervescent matrix.
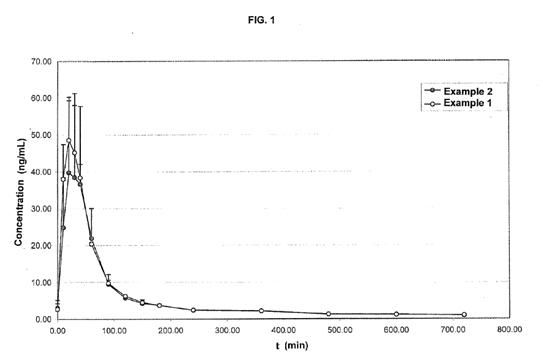
In the graph above it doesn’t appear much different from “Example 1”, however when you factor in the all important area under the curve (AUC) you would find a bioavailability increase of 30%. And this is why Advanced Muscle Science believes you will see a similar increase in bioavailability with the new line of effervescent matrix enhanced prohormones.

References1. Preparation and in vitro/in vivo characterization of porous sublingual tablets containing ternary kneaded solid system of vinpocetine with î-cyclodextrin and hydroxy Acid. Aburahma MH, El-Laithy HM, Hamza Yel-S. Cairo, Egypt : Sci Pharm., 2010, Vols. 78(2):363-79.
2. Lorenzo Bellorini, Luca Nocelli, Giorgio Zoppetti. PHARMACEUTICAL COMPOSITION FOR THE SUBLINGUAL ADMINISTRATION OF PROGESTERONE. US 2010/0240631 A1 United States, Oct 10, 2008.
3. Oral micronized progesterone. Bioavailability pharmacokinetics, pharmacological and therapeutic implications--a review. Sitruk-Ware R, Bricaire C, De Lignieres B, Yaneva H, Mauvais-Jarvis P. Paris, France. : Contraception., 1987, Vols. Oct;36(4):373-402.
