I know a lot of people were asking to see more information on our primary active in Vapor-XT. Here it is...feel free to post up questions/comments:
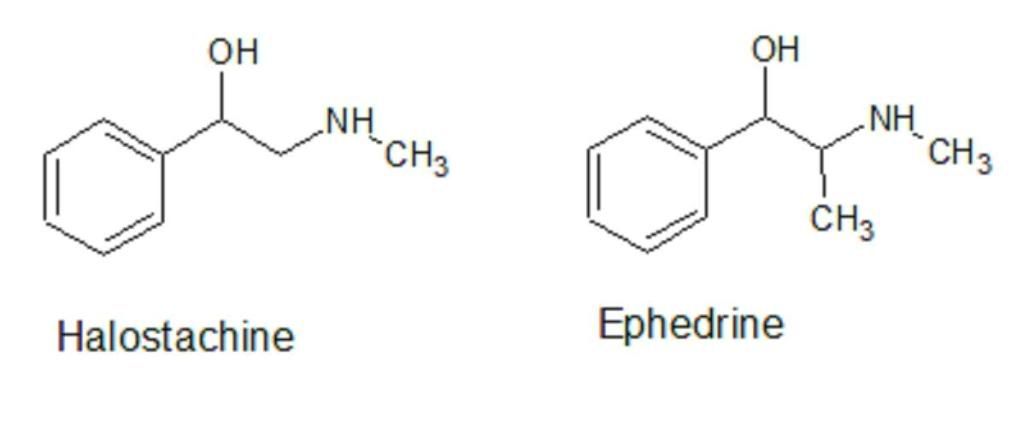
Halostachine, also known as N-methylphenylethanolamine, is a beta adrenergic agonist (1,2) similar in structure and activity to ephedrine that is found naturally in rye grass and is also produced exogenously in mammals and standardized from the plant Halostachys Caspica. Furthermore, it is a natural metabolite of phenylethylamine, which is found naturally in cocoa beans. Halostachine has pharmacological action similar to ephedrine but is slightly less potent and believed to be more safe (3). Halostachine is less toxic than ephedrine, with an LD50 twice that of ephedrine, 500 mg/kg ip vs 242 mg/kg sp which gives an indication of the relative toxicity of this ingredient to other similar products (4,5). Anecdotal testing of halostachine suggests that it is actually more effective than ephedrine, giving it a much better therapeutic index (efficacious dose)/(toxic dose). A paper by Shannn, Cone and Yousefnejad demonstrated no significant increases in heart rate with halostachine administration - even when administered intravenously at doses of several multiples of those proposed for supplemental use. This makes the favorable results more comparable to synephrine in effect(6). Increased heart rate and cardiovascular effect is a potential liability of ephedrine use and the main cause of the FDA action against ephedrine yet is reduced in synephrine, which is still on the market.
Like synephrine, the ease of production into methamphetamine is not much of a concern since halostachine has no methyl group in the side chain. Although we are not experts in illicit drug production, this critical difference should alleviate a major cause of ephedrine being banned in the US and other countries.
1. Liapakis G, Chan WC, Papadokostaki M, Javitch JA. Synergistic contributions of the functional groups of epinephrine to its affinity and efficacy at the beta2 adrenergic receptor. Mol Pharmacol. 2004 May;65(5):1181-90.
2. Fang Y, Ferrie AM. Label-free optical biosensor for ligand-directed functional selectivity acting on beta(2) adrenoceptor in living cells. FEBS Lett. 2008 Mar 5;582(5):558-64.
3. Manske & Holmes. The Alkaloids, Volume III. Chapter 22, 1953.
4. Halostachine MSDS
5. Ephedrine MSDS
6. Shannon HE, Cone EJ, Yousefnejad D. Physiologic effects and plasma kinetics of phenylethanolamine and its N-methyl homolog in the dog. J Pharmacol Exp Ther. 1981 May;217(2):379-85.
Halostachine, also known as N-methylphenylethanolamine, is a beta adrenergic agonist (1,2) similar in structure and activity to ephedrine that is found naturally in rye grass and is also produced exogenously in mammals and standardized from the plant Halostachys Caspica. Furthermore, it is a natural metabolite of phenylethylamine, which is found naturally in cocoa beans. Halostachine has pharmacological action similar to ephedrine but is slightly less potent and believed to be more safe (3). Halostachine is less toxic than ephedrine, with an LD50 twice that of ephedrine, 500 mg/kg ip vs 242 mg/kg sp which gives an indication of the relative toxicity of this ingredient to other similar products (4,5). Anecdotal testing of halostachine suggests that it is actually more effective than ephedrine, giving it a much better therapeutic index (efficacious dose)/(toxic dose). A paper by Shannn, Cone and Yousefnejad demonstrated no significant increases in heart rate with halostachine administration - even when administered intravenously at doses of several multiples of those proposed for supplemental use. This makes the favorable results more comparable to synephrine in effect(6). Increased heart rate and cardiovascular effect is a potential liability of ephedrine use and the main cause of the FDA action against ephedrine yet is reduced in synephrine, which is still on the market.
Like synephrine, the ease of production into methamphetamine is not much of a concern since halostachine has no methyl group in the side chain. Although we are not experts in illicit drug production, this critical difference should alleviate a major cause of ephedrine being banned in the US and other countries.

1. Liapakis G, Chan WC, Papadokostaki M, Javitch JA. Synergistic contributions of the functional groups of epinephrine to its affinity and efficacy at the beta2 adrenergic receptor. Mol Pharmacol. 2004 May;65(5):1181-90.
2. Fang Y, Ferrie AM. Label-free optical biosensor for ligand-directed functional selectivity acting on beta(2) adrenoceptor in living cells. FEBS Lett. 2008 Mar 5;582(5):558-64.
3. Manske & Holmes. The Alkaloids, Volume III. Chapter 22, 1953.
4. Halostachine MSDS
5. Ephedrine MSDS
6. Shannon HE, Cone EJ, Yousefnejad D. Physiologic effects and plasma kinetics of phenylethanolamine and its N-methyl homolog in the dog. J Pharmacol Exp Ther. 1981 May;217(2):379-85.

