So, I have been eatting more eggs lately and I feel good. I was just wondering what people have heard about eggs and cholesterol. I've heard it raises cholesteral and then I've read it really doesn't. My friend said he used to eat eggs everyday when he was working out and had no problem. He also said his dad eats eggs twice a day. What is your take on eggs and what have you heard about eggs and cholesterol. I just keep picturing that scene from Rocky where Stallone eats all those raw eggs.
natas9
New member
- Awards
- 0
eat em'
Cholesterol is much more hereditary than dietary. A couple of yokes aren't going to hurt. I wouldn't eat them raw though, the protein is much more bioavailable after cooking. Enjoy your eggs.
Cholesterol is much more hereditary than dietary. A couple of yokes aren't going to hurt. I wouldn't eat them raw though, the protein is much more bioavailable after cooking. Enjoy your eggs.
I heard the opposite - the bioavailability is higher when raw - you don't see foetus's cranking up the BBQ do you?Cholesterol is much more hereditary than dietary. A couple of yokes aren't going to hurt. I wouldn't eat them raw though, the protein is much more bioavailable after cooking. Enjoy your eggs.
If you have some info to back up your claim would be good.
Brother
Member
- Awards
- 0
Yea I heard the same.... that everything is better raw.. inculding eggs. Id rather have a tasty omlet anyday over a slimy cold egg. Im sure some foods are less likely to make you ill if theyve been cooked.
not that bad in protein shakes. Can't even taste it over the whey and milk.Yea I heard the same.... that everything is better raw.. inculding eggs. Id rather have a tasty omlet anyday over a slimy cold egg. Im sure some foods are less likely to make you ill if theyve been cooked.
yep - this is how I have them mainly. Whey, oats, milk, fruit - you can't taste or feel the egg texture.not that bad in protein shakes. Can't even taste it over the whey and milk.
Just buy pasteurized egg whites and add a cup to your regular workout shake. It tastes no different than adding some milk to your shake. And if your genetically predisposed to high cholesterol, you won't have to worry about it since the cholesterol is in the yolk.
the yolk also has protein, not to mention essential fatty acids!Just buy pasteurized egg whites and add a cup to your regular workout shake. It tastes no different than adding some milk to your shake. And if your genetically predisposed to high cholesterol, you won't have to worry about it since the cholesterol is in the yolk.
I found this article - should sort out a few myths...
The Incredible, Edible Egg Yolk
July, 2005
by Chris Masterjohn
Egg yolks? But they're full of cholesterol!
I'm sure you've heard it before. When you think of a "health freak," you don't think of someone eating egg yolks and discarding the white.
Think again.
Egg Nutrition: Yolk vs. White
Egg yolks are indeed full of cholesterol. Like most cholesterol-rich foods, they are jam-packed full of important nutrients, especially the fat-soluble vitamins and essential fatty acids.
In fact, the slew of nutrients in an egg yolk is so comprehensive that a few a day would offer better insurance than a multi-vitamin. Most importantly, the yolk contains most of the nutrients in an egg.
Egg whites, on the other hand, contain far fewer nutrients. The only thing that could justify their consumption is their attachment to their companion yolk.
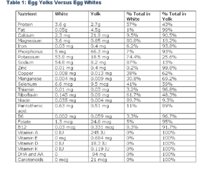
Don't believe it? Below is a table that compares the nutritional value of egg whites and yolks, with data provided by the USDA. I've included additional analysis in the last two columns that provides the percentage of the total nutrition found in the yolk and the percentage of total nutrition found in the white.
Data taken from the USDA Nutrient Database for Standard Reference, Release 15. AA and DHA data from NutritionData.Com. Since the article was written, the USDA has published revisions. The latest, Release 17, can be found here.
As you can see from the table, the yolk contains 100% of the carotenoids, essential fatty acids, vitamins A, E, D, and K (6 items). The white does not contain 100% of any nutrient.
The yolk contains more than 90% of the calcium, iron, phosphorus, zinc, thiamin, B6, folate, and B12, and 89% of the panthothenic acid (9 items). The white does not contain more than 90% of any nutrient, but contains over 80% of the magnesium, sodium, and niacin (3 items).
The yolk contains between 50% and 80% of the copper, manganese, and selenium, while the white contains between 50% and 80% of the potassium, riboflavin, and protein.
It should also be kept in mind that the yolk of an egg is smaller than the white. Where the white contains a slim majority of nutrients, such as protein, this is not due to a greater concentration in the white, but simply to the fact that there is more white in the egg than yolk.
Egg Yolks Would Resolve Americans' Most Common Nutrient Deficiencies
According to the Executive Summary of the Third Report on Nutrition Monitoring in the United States by the Interagency Board for Nutrition Monitoring and Related Research of the Federation of American Societies for Experimental Biology's Life Sciences Research Office, the following is true:
Most groups have a deficient median intake of magnesium.
Several groups have a deficient median intake of calcium.
Children aged 1-2 and most groups of females have a deficient median intake of iron.
Blacks over the age of 16 and Mexican-Americans over the age of 60 have a deficient median intake of folate.
All age groups and races have a deficient median intake of vitamins A, E, B6, and copper.
Considering this information, the importance of the egg yolk and relative unimportance of the egg white becomes even more clear. The yolk contains the majority of the copper, nearly all of the calcium, iron, folate, and B6, and 100% of the vitamins A and E.
The white, on the other hand, is only useful as an added source of magnesium, or if the diet is on the whole deficient in protein. The simple addition of an adequate amount of meat in the diet would provide for both.
Finally, eggs are an excellent source of carotenoids. These are primarily highly absorbable forms of lutein and its partner zeaxanthin. These carotenoids accumulate in the back of the eye and appear to protect against age-related macular degeneration. There is no RDA for them, as researchers are still trying to understand their importance. All of the lutein and zeaxanthin in an egg is contained in the yolk.
Egg Yolks Contain Essential Fatty Acids DHA and Arachidonic Acid
One important set of nutrients that should not be overlooked is the long-chain essential fatty acids. Egg yolks contain the long-chain omega-3 fatty acid DHA, which is necessary for the brain and proper retinal function in the eye, and the long-chain omega-6 fatty acid arachidonic acid, which is required for the healthy skin, hair, libido, reproduction, growth and response to injury. These fatty acids are primarily needed by young children, pregnant and lactating women, and people with degenerative diseases involving oxidative stress, especially those of the nervous system such as Alzheimer's. While fatty fish and cod liver oil supply DHA in larger amounts, egg yolks have an advantage over these foods because they also contain arachidonic acid and because they do not contain EPA, which interferes with arachidonic acid metabolism.
According to NutritionData.Com, one egg yolk contains 75 mg of arachidonic acid (AA), 20 mg of DHA, but no EPA. As I describe in my Special Report, How Essential Are the Essential Fatty Acids?, DHA and AA are the two fatty acids essential to humans and other mammals, while EPA interferes with the body's use of AA and probably does not belong in the mammalian body at all.
Animal foods from animals raised on pasture are likely much richer in DHA. In all eggs, both the DHA and AA are contained in the yolk.
To Cook, or Not to Cook? The Benefits of Raw Egg Yolks
Many people believe that the health benefits of egg yolks are greater when the yolks are consumed raw. Heat destroys enzymes, reduces the amounts of certain nutrients, and may make the amino acid cysteine less available, which is needed to synthesize the master antioxidant of the cell, glutathione.
Those who eat raw egg yolks report easier digestion, increased stamina, and resistance to illness — not to mention a quicker snack if they're on-the-go.
That said, there is little evidence beyond such anecdotes that egg yolks are truly more beneficial when consumed raw.
There is also little evidence to support the common belief that consuming raw egg yolks is dangerous. Please see Dr. Mercola's article on the safety of eating raw egg yolks if you are concerned about this.
Raw Egg Whites Contain Digestive Enzyme Inhibitors and Anti-Nutrients
Raw egg whites should not be consumed. They contain inhibitors of the digestive enzyme trypsin, which are destroyed by heat. Consuming 100 grams of raw egg white with one egg yolk compared to consuming the same food cooked was shown in one study to reduce protein digestion from 90 percent down to 50 percent.
Raw egg whites also contain an anti-nutrient called avidin. Avidin is a glycoprotein that binds to the B vitamin biotin, preventing its absorption. Biotin is necessary for fatty acid synthesis and the maintenance of blood sugar, and is especially important during pregnancy when biotin status declines.
Residual Egg White Avidin — Cooking Does Not Fully Destroy the Anti-Nutrients
It is a myth that light cooking completely destroys the avidin.
According to this study, poaching eggs only destroys one third of the avidin while even frying leaves 30 percent of it behind.
This raises the question of whether there is a net nutritional advantage to eating any egg whites at all. Most likely, it depends on the individual person. There is controversy over whether biotin produced in the intestinal tract is absorbed — if intenstinal biotin production is indeed nutritionally important, then people whose intestinal flora are less avid producers of biotin probably need to be more concerned about the potential adverse effects of consuming egg white.
Finding The Right Kind of Eggs
Pastured eggs, meaning eggs from chickens that are free to forage for grass and insects, are of much higher nutritional quality than eggs from confinement chickens. The marginal increase in value, of course, is found mostly in the yolk.
Insects provide a higher DHA content, found exclusive in the yolk, and grass provides a higher vitamin E and carotene content, also found exclusively in the yolk. Egg yolks from pastured chickens are thus a powerful supplement to a healthy diet — a super-food — providing necessary nutrients in which the Standard American Diet is deficient.
To find a source of eggs from chickens raised on pasture, you can visit LocalHarvest.org and do a search for "eggs pastured" or "eggs grass fed" with your zip code. You can also visit EatWild.com and click on your state for a list of farms that pasture their animals.
Additionally, you may be able to find roadside stands in your area that sell eggs from pastured chickens. Be sure to inquire about the farming practices, to make sure that the chickens are able to forage for both grass and insects.
Back to the Basics: Taste!
The truth is that most satisfying meals one could make with eggs just don't taste right without both the yolk and the white. Most baked goods come out with a richer taste and a better texture when the yolks are included. Food should provide good nutrition — for which inclusion of the yolks is necessary! — but it should also taste good.
Food should be fun. It should be rewarding to cook, delicious to eat, and relaxing to indulge in.
The anti-cholesterol establishment upholds its poor theory and unjustified conclusions only to condemn us to a bland and unsatisfying diet, the cornerstone of which is "light cooking" with bland and taste-challenged "foods" like the notorious, emasculated, yolkless egg white. Fear not.
You are now armed with the raw facts from the USDA's nutrition database that shows that missing out on the egg yolks means missing out on the nutrition in your breakfast. Take heart in this the next time you enjoy the incredible, edible egg yolk.
The Incredible, Edible Egg Yolk
July, 2005
by Chris Masterjohn
Egg yolks? But they're full of cholesterol!
I'm sure you've heard it before. When you think of a "health freak," you don't think of someone eating egg yolks and discarding the white.
Think again.
Egg Nutrition: Yolk vs. White
Egg yolks are indeed full of cholesterol. Like most cholesterol-rich foods, they are jam-packed full of important nutrients, especially the fat-soluble vitamins and essential fatty acids.
In fact, the slew of nutrients in an egg yolk is so comprehensive that a few a day would offer better insurance than a multi-vitamin. Most importantly, the yolk contains most of the nutrients in an egg.
Egg whites, on the other hand, contain far fewer nutrients. The only thing that could justify their consumption is their attachment to their companion yolk.
Don't believe it? Below is a table that compares the nutritional value of egg whites and yolks, with data provided by the USDA. I've included additional analysis in the last two columns that provides the percentage of the total nutrition found in the yolk and the percentage of total nutrition found in the white.
Data taken from the USDA Nutrient Database for Standard Reference, Release 15. AA and DHA data from NutritionData.Com. Since the article was written, the USDA has published revisions. The latest, Release 17, can be found here.
As you can see from the table, the yolk contains 100% of the carotenoids, essential fatty acids, vitamins A, E, D, and K (6 items). The white does not contain 100% of any nutrient.
The yolk contains more than 90% of the calcium, iron, phosphorus, zinc, thiamin, B6, folate, and B12, and 89% of the panthothenic acid (9 items). The white does not contain more than 90% of any nutrient, but contains over 80% of the magnesium, sodium, and niacin (3 items).
The yolk contains between 50% and 80% of the copper, manganese, and selenium, while the white contains between 50% and 80% of the potassium, riboflavin, and protein.
It should also be kept in mind that the yolk of an egg is smaller than the white. Where the white contains a slim majority of nutrients, such as protein, this is not due to a greater concentration in the white, but simply to the fact that there is more white in the egg than yolk.
Egg Yolks Would Resolve Americans' Most Common Nutrient Deficiencies
According to the Executive Summary of the Third Report on Nutrition Monitoring in the United States by the Interagency Board for Nutrition Monitoring and Related Research of the Federation of American Societies for Experimental Biology's Life Sciences Research Office, the following is true:
Most groups have a deficient median intake of magnesium.
Several groups have a deficient median intake of calcium.
Children aged 1-2 and most groups of females have a deficient median intake of iron.
Blacks over the age of 16 and Mexican-Americans over the age of 60 have a deficient median intake of folate.
All age groups and races have a deficient median intake of vitamins A, E, B6, and copper.
Considering this information, the importance of the egg yolk and relative unimportance of the egg white becomes even more clear. The yolk contains the majority of the copper, nearly all of the calcium, iron, folate, and B6, and 100% of the vitamins A and E.
The white, on the other hand, is only useful as an added source of magnesium, or if the diet is on the whole deficient in protein. The simple addition of an adequate amount of meat in the diet would provide for both.
Finally, eggs are an excellent source of carotenoids. These are primarily highly absorbable forms of lutein and its partner zeaxanthin. These carotenoids accumulate in the back of the eye and appear to protect against age-related macular degeneration. There is no RDA for them, as researchers are still trying to understand their importance. All of the lutein and zeaxanthin in an egg is contained in the yolk.
Egg Yolks Contain Essential Fatty Acids DHA and Arachidonic Acid
One important set of nutrients that should not be overlooked is the long-chain essential fatty acids. Egg yolks contain the long-chain omega-3 fatty acid DHA, which is necessary for the brain and proper retinal function in the eye, and the long-chain omega-6 fatty acid arachidonic acid, which is required for the healthy skin, hair, libido, reproduction, growth and response to injury. These fatty acids are primarily needed by young children, pregnant and lactating women, and people with degenerative diseases involving oxidative stress, especially those of the nervous system such as Alzheimer's. While fatty fish and cod liver oil supply DHA in larger amounts, egg yolks have an advantage over these foods because they also contain arachidonic acid and because they do not contain EPA, which interferes with arachidonic acid metabolism.
According to NutritionData.Com, one egg yolk contains 75 mg of arachidonic acid (AA), 20 mg of DHA, but no EPA. As I describe in my Special Report, How Essential Are the Essential Fatty Acids?, DHA and AA are the two fatty acids essential to humans and other mammals, while EPA interferes with the body's use of AA and probably does not belong in the mammalian body at all.
Animal foods from animals raised on pasture are likely much richer in DHA. In all eggs, both the DHA and AA are contained in the yolk.
To Cook, or Not to Cook? The Benefits of Raw Egg Yolks
Many people believe that the health benefits of egg yolks are greater when the yolks are consumed raw. Heat destroys enzymes, reduces the amounts of certain nutrients, and may make the amino acid cysteine less available, which is needed to synthesize the master antioxidant of the cell, glutathione.
Those who eat raw egg yolks report easier digestion, increased stamina, and resistance to illness — not to mention a quicker snack if they're on-the-go.
That said, there is little evidence beyond such anecdotes that egg yolks are truly more beneficial when consumed raw.
There is also little evidence to support the common belief that consuming raw egg yolks is dangerous. Please see Dr. Mercola's article on the safety of eating raw egg yolks if you are concerned about this.
Raw Egg Whites Contain Digestive Enzyme Inhibitors and Anti-Nutrients
Raw egg whites should not be consumed. They contain inhibitors of the digestive enzyme trypsin, which are destroyed by heat. Consuming 100 grams of raw egg white with one egg yolk compared to consuming the same food cooked was shown in one study to reduce protein digestion from 90 percent down to 50 percent.
Raw egg whites also contain an anti-nutrient called avidin. Avidin is a glycoprotein that binds to the B vitamin biotin, preventing its absorption. Biotin is necessary for fatty acid synthesis and the maintenance of blood sugar, and is especially important during pregnancy when biotin status declines.
Residual Egg White Avidin — Cooking Does Not Fully Destroy the Anti-Nutrients
It is a myth that light cooking completely destroys the avidin.
According to this study, poaching eggs only destroys one third of the avidin while even frying leaves 30 percent of it behind.
This raises the question of whether there is a net nutritional advantage to eating any egg whites at all. Most likely, it depends on the individual person. There is controversy over whether biotin produced in the intestinal tract is absorbed — if intenstinal biotin production is indeed nutritionally important, then people whose intestinal flora are less avid producers of biotin probably need to be more concerned about the potential adverse effects of consuming egg white.
Finding The Right Kind of Eggs
Pastured eggs, meaning eggs from chickens that are free to forage for grass and insects, are of much higher nutritional quality than eggs from confinement chickens. The marginal increase in value, of course, is found mostly in the yolk.
Insects provide a higher DHA content, found exclusive in the yolk, and grass provides a higher vitamin E and carotene content, also found exclusively in the yolk. Egg yolks from pastured chickens are thus a powerful supplement to a healthy diet — a super-food — providing necessary nutrients in which the Standard American Diet is deficient.
To find a source of eggs from chickens raised on pasture, you can visit LocalHarvest.org and do a search for "eggs pastured" or "eggs grass fed" with your zip code. You can also visit EatWild.com and click on your state for a list of farms that pasture their animals.
Additionally, you may be able to find roadside stands in your area that sell eggs from pastured chickens. Be sure to inquire about the farming practices, to make sure that the chickens are able to forage for both grass and insects.
Back to the Basics: Taste!
The truth is that most satisfying meals one could make with eggs just don't taste right without both the yolk and the white. Most baked goods come out with a richer taste and a better texture when the yolks are included. Food should provide good nutrition — for which inclusion of the yolks is necessary! — but it should also taste good.
Food should be fun. It should be rewarding to cook, delicious to eat, and relaxing to indulge in.
The anti-cholesterol establishment upholds its poor theory and unjustified conclusions only to condemn us to a bland and unsatisfying diet, the cornerstone of which is "light cooking" with bland and taste-challenged "foods" like the notorious, emasculated, yolkless egg white. Fear not.
You are now armed with the raw facts from the USDA's nutrition database that shows that missing out on the egg yolks means missing out on the nutrition in your breakfast. Take heart in this the next time you enjoy the incredible, edible egg yolk.
Attachments
-
172.8 KB Views: 608
Just eat the whole egg!
If you already have a prexisting problem with your cholesterol, i think you have somethings else to worry about/ change than egg yolks
If you already have a prexisting problem with your cholesterol, i think you have somethings else to worry about/ change than egg yolks
My cholesteral was 156 about 5-6 years ago before I started working out... I've been eating eggs almost every day for the past 5 years (4-5 whole eggs)... My cholesteral was checked last week and was 157.... So take that as it is, I don't think eating eggs does crap to your cholesteral.... Hereditary plays a bigger role.
Raw poultry, FTW!
The cholesterol in eggs is mostly HDL! That's the GOOD kind.
What he said____^^^The cholesterol in eggs is mostly HDL! That's the GOOD kind.
I don't know if it's in my head, but when I eat eggs I do have more energy. Thanks for the article.
My pre bed meal has 4 egg whites, and one egg yolk. I try to stay away from the yolks.
natas9
New member
- Awards
- 0
I heard the opposite - the bioavailability is higher when raw - you don't see foetus's cranking up the BBQ do you?
If you have some info to back up your claim would be good.
The Journal of Nutrition Vol. 128 No. 10 October 1998, pp. 1716-1722
Digestibility of Cooked and Raw Egg Protein in Humans as Assessed by Stable Isotope Techniques1,2,3
Pieter Evenepoel, Benny Geypens, Anja Luypaerts, Martin Hiele, Yvo Ghoos4, and Paul Rutgeerts
Department of Medicine, Division of Gastroenterology and Gastrointestinal Research Centre, University Hospital Leuven, B-3000 Leuven, Belgium
Egg proteins contribute substantially to the daily nitrogen allowances in Western countries and are generally considered to be highly digestible. However, information is lacking on the true ileal digestibility of either raw or cooked egg protein. The recent availability of stable isotope-labeled egg protein allowed determination of the true ileal digestibility of egg protein by means of noninvasive tracer techniques. Five ileostomy patients were studied, once after ingestion of a test meal consisting of 25 g of cooked 13C- and 15N-labeled egg protein, and once after ingestion of the same test meal in raw form. Ileal effluents and breath samples were collected at regular intervals after consumption of the test meal and analyzed for 15N- and 13C-content, respectively. The true ileal digestibility of cooked and raw egg protein amounted to 90.9 ± 0.8 and 51.3 ± 9.8%, respectively. A significant negative correlation (r = 0.92, P < 0.001) was found between the 13C-recovery in breath and the recovery of exogenous N in the ileal effluents. In summary, using the 15N-dilution technique we demonstrated that the assimilation of cooked egg protein is efficient, albeit incomplete, and that the true ileal digestibility of egg protein is significantly enhanced by heat-pretreatment. A simple 13C-breath test technique furthermore proved to be a suitable alternative for the evaluation of the true ileal digestibility of egg protein.
Other approaches therefore have been developed to distinguish the exogenous from the endogenous protein fraction in the intestinal contents including the use of dietary protein labeled with homoarginine (Siriwan et al. 1994) and the evaluation of the endogenous/exogenous ratio of effluents, which is extrapolated by comparing the amino acid composition of both the chyme and the meal (Baglieri et al. 1995, Mahé et al. 1994b). Primarily for safety reasons, however, the tracer dilution technique using stable isotopes is the most appropriate for studies involving human subjects. The widespread use of this technique for the study of protein digestibility has been hindered for a long time by the lack of an adequate substrate, i.e., stable isotope-labeled protein. In recent years, different proteins labeled with 15N have become available (e.g., milk protein, soy protein or pea protein) (Gausserès et al. 1996, Kayser et al. 1992, Mahé et al. 1994b). Very recently, we succeeded in producing both 15N- and 13C-labeled egg protein in an easily reproducible and highly efficient manner (Evenepoel et al. 1997).
The digestibility of protein is affected by its state of processing before ingestion. Both an enhanced and a reduced protein digestibility have been observed after food processing (e.g., boiling, drying, deep-freezing or microwave-heating) (Öste 1991). Using the "ileostomy model," this study attempted the following: 1) to determine the amount of nitrogen escaping digestion and absorption after ingestion of a physiologic load of egg protein and to distinguish the exogenous from the endogenous fraction; 2) to validate the 13C-egg protein breath test as a tool for the evaluation of protein digestibility in vivo; and 3) to evaluate the influence of heat-pretreatment on the efficiency of egg protein assimilation. Protein assimilation is defined as the overall process of digestion and absorption finally resulting in the appearance of free amino acids in the portal circulation.
Protein test meal. The protein test meal consisted of 100 g of 13C-labeled egg white, 100 g of 15N-labeled egg white (markers of protein assimilation) and the yolk of one egg doped with 74 kBq of [1-14C]octanoic acid (Dupont, NEN Research, Boston, MA) (marker of gastric emptying) (Ghoos et al. 1993). Five microcurie 3H-polyethylene glycol (3H-PEC) 4000 was added to the test meal as a nonabsorbable radiolabeled transit marker. All constituents were homogenized before ingestion. The test meal had to be consumed either raw or after being cooked in a microwave oven (see Experimental design). The methodology for obtaining large amounts of highly enriched egg proteins labeled with stable isotopes was extensively described elsewhere (Evenepoel et al. 1997). Briefly, 13C- or 15N-labeled proteins were produced by giving laying hens free access to a food containing 25% of the (NRC required) leucine content as free [1-13C]leucine (99 mol%, Euriso-top, Saint-Aubin, France) and [15N]leucine (99 mol%, Euriso-top), respectively. The yolk and egg white fractions of the enriched eggs were separated and pooled. The isotopic enrichment of both pools was determined using a continuous flow elemental analyzer isotope ratio mass spectrometer (IRMS)5 (ANCA-SL, Europa Scientific, Crewe, UK). Knowing the exact amino acid composition and the isotopic enrichment of the egg white, the amount of [1-13C]leucine (99 mol%) incorporated could be calculated. This value was taken into account in the calculation procedures, outlined below. Because "redistribution" of the 15N-label is likely to occur in the hen via transamination, the 15N-labeled egg protein can be assumed to be uniformly labeled. Total caloric content of the test meal was 624 kJ (25 g protein, 5.56 g fat and a negligible amount of carbohydrate).
Experimental design. Each subject was studied in two different randomly applied test situations as follows: 1) after ingestion of the cooked egg protein test meal ("cooked"), and 2) after ingestion of the same but raw protein test meal ("raw"). The test situations were separated from each other by a time span of at least 1 wk. Measurements of gastric emptying (breath test technique), small intestinal transit time (nonabsorbable transit marker) and protein assimilation (both by breath test technique and analysis of ileostomy effluents) were performed on both occasions.
All subjects were studied after an overnight fast of at least 12 h. At 0845 h on the test day, they were asked to consume the meal together with 200 mL of water within 15 min. Sodium chloride seasoning was allowed. No further food was eaten until 1500 h when the patients ingested 200 mL of a formula food, based on whey, casein and lactalbumin (Nutridrink, Nutricia, Zoetermeer, The Netherlands). Five milligrams of phenol red was added to this formula food as a nonabsorbable transit marker. Drinking of water was permitted from 1200 h on.
The patients emptied their ileostomy bag immediately after ingestion of the meal (i.e., at 0900 h), at 1-h intervals throughout the day until 1900 h and at 0900 h the next morning. Breath samples for 13CO2 and 14CO2 were collected before ingestion of the meal and every 15 min thereafter for 6 h. The experimental design is schematically represented in Figure 1.
Breath test results. As shown in Figure 2, the course of both the 13CO2 (panel A) and 14CO2 (panel B) excretion rate was influenced by the state of the test meal, whether cooked or raw. The parameters of protein assimilation and gastric emptying obtained in both test situations are given in Table 1. Significant differences between the two test situations were found for several variables.
Fig 2. Mean 13CO2 (A, representing protein assimilation) and 14CO2 (B, representing gastric emptying) excretion rates, expressed as percentage of the administered dose of 13/14C excreted per h (% dose/h) in ileostomy patients after ingestion of a test meal consisting of 25 g of either cooked or raw egg protein. Values are means ± SEM, n = 5.
Table 1. Protein assimilation, gastric emptying and ileal emptying and the small bowel transit time in ileostomy patients after ingestion of cooked and raw test meals consisting of 25 g egg protein1
Ileostomy effluents.
Validation of use of 3H-PEG as transit marker. There was a significant correlation (r2 > 0.97, P < 0.0001) between the delivery of the radioactive marker in the ileostomy effluent and the delivery of exogenous protein in each test condition (Fig. 3).
Profile of delivery of meal residues from the ileum. Ileal emptying of the test meal began in the first hour after ingestion of the cooked and raw test meals (Fig. 4). The emptying rate accelerated temporarily in the 1- to 2-h period and clearly peaked in the 6- to 7-h period. Fifty percent of the meal had emptied from the ileostomy (ileal t1/2) by 5.33 ± 0.76 and 5.29 ± 0.50 h after the ingestion of the cooked meal and raw test meal, respectively (Table 1).
Fig 4. Ileal emptying profile of the cooked and raw protein meal, expressed in percentage of administered dose of 3H-polyethyle glycol (PEG) recovered in the effluents of ileostomy patients per hour (% dose 3H-PEG/h). Values are means ± SEM, n = 5.
Exogenous and endogenous nitrogen in ileostomy effluents. Figure 5 shows the endogenous and exogenous nitrogen fraction profiles after 15N-egg protein ingestion in the two test conditions. The cumulative quantities of exogenous and endogenous nitrogen, recovered in the ileostomy effluent over 24 h are given in Table 2. A significantly greater amount of exogenous nitrogen was recovered in the ileal effluent over 24 h after ingestion of the raw test meal compared with the cooked test meal (1949.4 ± 390.3 m vs. 360.6 ± 30.6 mg, P < 0.05). Taking into account these data and the amount of N ingested, it was possible to calculate the true ileal digestibility of egg protein, i.e., the percentage of exogenous egg protein assimilated in the small intestine. The true ileal digestibility of raw egg protein was significantly impaired compared with that of cooked egg protein (51.3 ± 9.8 vs. 90.9 ± 0.8%, P < 0.05).
Fig 5. Delivery of endogenous (Nendo), exogenous (Nexo) and total (Ntot) nitrogen in ileal effluents of ileostomy patients after ingestion of cooked and raw test meals consisting of 25 g egg protein (=4000 mg N). Values are means ± SEM, n = 5.
View this table:
Table 2. Exogenous and endogenous nitrogen yield over 24 h in ileal effluents of ileostomy patients after ingestion of 25 g of either cooked or raw 15N-labeled egg protein1,2
Correlations. A significant negative correlation was found between the amount of exogenous N recovered in the ileostomy effluent and the cumulative percentage administered dose of 13C recovered in breath over 6 h (Fig. 6). There were no significant correlations between the gastric emptying half time (gastric t1/2) and the small bowel transit time (defined as ileal t1/2 gastric t1/2), nor between the small bowel transit time and the true ileal digestibility of egg protein in each of the test conditions.
Fig 6. Correlation between the cumulative amount of exogenous nitrogen, recovered in the ileal effluents over 24 h (Nexo 24 h cum) and the cumulative percentage of administered dose of 13C, recovered in breath over 6 h (% dose 13C cum 6 h) in ileostomy patients.
Dietary protein provides 10-15% of the total energy intake of healthy subjects consuming a standard Western diet and supplies the essential amino acids required for protein synthesis. The nutritional value of dietary protein for humans is usually determined from a "chemical score" or more recently from the "protein digestibility-corrected amino acid score" in which digestibility represents an important aspect (Young and Pellett 1991). According to results obtained from nitrogen balance studies in healthy subjects, the true (fecal) digestibility of (spray dried whole) egg protein has been estimated to be as high as 92-97% (Bodwell et al. 1980). However, because nitrogen is intensely metabolized, absorbed and secreted in the colon, fecal digestibility values do not necessarily equal ileal digestibility values. This has been demonstrated in an animal study, in which fecal and ileal digestibility were measured simultaneously (Mosenthin et al. 1994). Because the ileal digestibility represents the efficiency of protein assimilation, ileal digestibility values are more relevant from a nutritional point of view than are fecal digestibility values. The ileal digestibility of different protein sources has already been assessed in healthy volunteers with the use of an intestinal perfusion technique (Chung et al. 1979, Gausserès et al. 1996, Mahé et al. 1994a). This technique, however, is time-consuming and invasive and, moreover, appears to delay gastric emptying and to shorten transit time in the small intestine (Read et al. 1983). Shortening of the transit time may compromise the assimilation of macronutrients (Chapman et al. 1985, Holgate and Read 1993). Because ileal effluents are easily collectible in ileostomy patients, nitrogen balance studies in these subjects are an attractive alternative for the evaluation of ileal digestibility (Chapman et al. 1985, Fuller et al. 1994, Gibson et al. 1976, Rowan et al. 1994, Sandström et al. 1986). A common problem encountered in studies designed to determine the ileal digestibility is the differentiation between the exogenous and endogenous origin of nitrogen recovered in ileal effluents. Because we recently obtained large amounts of highly enriched 15N-labeled egg proteins (Evenepoel et al. 1997), we were able to apply the isotope dilution technique for the assessment of the true ileal digestibility of egg protein.
Five healthy ileostomy patients were studied after ingestion of a physiologic load (25 g) of egg protein, labeled with 13C and 15N. The double labeling of the test meal allowed the evaluation of egg protein assimilation by both 13C-breath test technique and direct analysis of the ileostomy effluents (15N-dilution technique). The subjects were studied in different experimental conditions to assess the influence of heat-pretreatment on the assimilation efficiency of egg protein.
After ingestion of the cooked egg protein meal, a substantial quantity of nitrogen was recovered in the ileal effluent over 24 h. The calculated yield of endogenous nitrogen (i.e., 0.40 g N) was close to the yield of 0.55 g N obtained by other researchers after ingestion of 17 g of pea protein (Gausserès et al. 1994). The calculated true ileal digestibility of cooked egg protein amounted to 91%. This finding demonstrates that even cooked egg protein, which has generally been considered to be easily digestible, is malabsorbed to some extent after ingestion of a physiologic load. Incomplete assimilation of dietary protein may have important consequences not only from a nutritional point of view, but also from a gastrointestinal point of view. Indeed, some metabolites resulting from bacterial fermentation of malabsorbed proteins in the colon have been implicated in the ethiopathogenesis of diseases such as colonic cancer and ulcerative colitis (Macfarlane and Cummings 1991, Pitcher and Cummings 1994, Visek 1978). It has already been reported extensively that food processing can influence protein digestibility both beneficially and detrimentally (Öste 1991). Egg white protein is generally considered to be less digestible than heat-pretreated egg white protein. However, no data are available concerning the magnitude of this impairment in vivo. In this study, it was shown that after ingestion of 25 g of raw egg protein, almost 50% is malabsorbed over 24 h. The higher digestibility of cooked egg protein presumably results from structural changes in the protein molecule induced by heating, thereby enabling the digestive enzymes to gain broader access to the peptide bonds. It has been suggested that the reduced digestibility of raw egg white is at least partially related to the presence of trypsin inhibitors in raw egg white (Matthews 1990). Ovomucoid is quantitatively the most important trypsin inhibitor (Gilbert 1971, Kassell 1970). Ovomucoid, however, does not react with human trypsin and, moreover, is relatively heat stable (Kasell 1970). Whether other egg trypsin inhibitors (e.g., ovoinhibitor or papain inhibitor) interfere with the digestibility of unprocessed egg white protein is unknown.
Interestingly, the yield of endogenous nitrogen after ingestion of the raw protein meal (i.e., 0.2 g N) was significantly lower compared with the cooked protein meal. This finding is in accordance with a recent study in which it was demonstrated that undigested protein, in contrast to digested protein, only weakly stimulates gallbladder emptying and pancreatic enzyme secretions (Thimister et al. 1996).
Labeling of the test meal with 14C-octanoic acid allowed us to follow gastric emptying simultaneously. The raw test meal was emptied significantly more quickly than the cooked test meal, most probably because of its liquid consistency. Small intestinal transit of the raw test meal, on the other hand, tended to be slower than that of the cooked test meal. A shortened transit time therefore cannot account for the observed decreased digestibility of raw egg protein.
In this study, a significant negative correlation was observed between the percentage of administered dose 13C recovered in breath over 6 h and the amount of exogenous nitrogen recovered in the ileostomy effluent over 24 h. This finding implies that the 13C-egg protein breath test may be regarded as an accurate technique for the evaluation of protein digestibility. Although the 13C-egg protein breath test is only semiquantitative (i.e., it reveals adequacy of function compared with a normal standard), it has many advantages (i.e., it is noninvasive, simple and reproducible), by which it yet might be an attractive alternative for the evaluation of the efficiency of protein assimilation in healthy volunteers in different experimental conditions as well as in patients with digestive diseases. Moreover, the information obtained with breath tests represents a dynamic evaluation, rather than a static estimation.
No significant correlation was found between the gastric t1/2 and the small bowel transit time, which is in agreement with other studies (Read et al. 1982 and 1986). These results, however, should be interpreted with caution because of the small number of subjects studied.
In conclusion, with the use of stable isotope techniques, we were able to determine the amounts of egg protein escaping digestion and absorption in the small intestine after ingestion of a physiologic load. Native egg protein is malabsorbed to an important extent. The assimilation of egg protein is facilitated by heat-pretreatment, but remains incomplete. The excellent correlation found between digestibility values and 13C-breath test data validated the 13C-egg protein breath test as an accurate alternative for the evaluation of protein digestibility. Because of its many advantages, the 13C-egg protein breath test might be very interesting for the study of protein digestibility under different experimental conditions.
--------------------------------------------------------------------------------
natas9
New member
- Awards
- 0
In short:
...In conclusion, with the use of stable isotope techniques, we were able to determine the amounts of egg protein escaping digestion and absorption in the small intestine after ingestion of a physiologic load. Native egg protein is malabsorbed to an important extent. The assimilation of egg protein is facilitated by heat-pretreatment, but remains incomplete. The excellent correlation found between digestibility values and 13C-breath test data validated the 13C-egg protein breath test as an acurate alternative for the evaluation of protein digestibilty.....the true ileal digestibility of cooked and raw egg protein amounted to 90.0+/-0.8 and 51.3+/-9.8%, respectively....the true ileal digestibility of egg protein is significantly enhanced by heat-pretreatment...
91% bio-available cooked/51% bio-available raw.
Pasteurized eggs in a carton are not a bad option since pasteurization involves heating.
...In conclusion, with the use of stable isotope techniques, we were able to determine the amounts of egg protein escaping digestion and absorption in the small intestine after ingestion of a physiologic load. Native egg protein is malabsorbed to an important extent. The assimilation of egg protein is facilitated by heat-pretreatment, but remains incomplete. The excellent correlation found between digestibility values and 13C-breath test data validated the 13C-egg protein breath test as an acurate alternative for the evaluation of protein digestibilty.....the true ileal digestibility of cooked and raw egg protein amounted to 90.0+/-0.8 and 51.3+/-9.8%, respectively....the true ileal digestibility of egg protein is significantly enhanced by heat-pretreatment...
91% bio-available cooked/51% bio-available raw.
Pasteurized eggs in a carton are not a bad option since pasteurization involves heating.
I don't think I could live with myself throwing away perfectly good egg yolksMy pre bed meal has 4 egg whites, and one egg yolk. I try to stay away from the yolks.
That study was done on digestibility, not bioavailibility. Your summary should say 91% digestible cooked/51% digestible raw. Digestibility and bioavailability are not one in the same.In short:
...In conclusion, with the use of stable isotope techniques, we were able to determine the amounts of egg protein escaping digestion and absorption in the small intestine after ingestion of a physiologic load. Native egg protein is malabsorbed to an important extent. The assimilation of egg protein is facilitated by heat-pretreatment, but remains incomplete. The excellent correlation found between digestibility values and 13C-breath test data validated the 13C-egg protein breath test as an acurate alternative for the evaluation of protein digestibilty.....the true ileal digestibility of cooked and raw egg protein amounted to 90.0+/-0.8 and 51.3+/-9.8%, respectively....the true ileal digestibility of egg protein is significantly enhanced by heat-pretreatment...
91% bio-available cooked/51% bio-available raw.
Pasteurized eggs in a carton are not a bad option since pasteurization involves heating.
natas9
New member
- Awards
- 0
That study was done on digestibility, not bioavailibility. Your summary should say 91% digestible cooked/51% digestible raw. Digestibility and bioavailability are not one in the same
Your splitting hairs here. The study specifically addresses egg protein absorption in the body. If your body can only digest raw eggs efficiently at 51%, their bioavailability means little when compared to 91% for cooked eggs.
Now, if you're questioning the bioavailability of eggs...that's a different story.
TouchingCloth
New member
- Awards
- 0
When that article mentioned Dr. Mercola as a source for raw eggs I turned off.
I eat 8-10 whole eggs a day, and my cholesterol is good.
I eat 8-10 whole eggs a day, and my cholesterol is good.
Damitri
New member
- Awards
- 0
Usually I just eat the whites
ILiftBig
Member
- Awards
- 0
Eggs are high in cholesterol in fact one large egg has about 213 milligrams (mg) of cholesterol — all of which is found in the yolk. We also suspect that a diet high in cholesterol can contribute to elevated blood cholesterol levels. However, the extent to which dietary cholesterol raises blood cholesterol levels isn't clear. Many scientists believe that saturated fats and trans fats have a greater impact than does dietary cholesterol in raising blood cholesterol.So, I have been eatting more eggs lately and I feel good. I was just wondering what people have heard about eggs and cholesterol. I've heard it raises cholesteral and then I've read it really doesn't. My friend said he used to eat eggs everyday when he was working out and had no problem. He also said his dad eats eggs twice a day. What is your take on eggs and what have you heard about eggs and cholesterol. I just keep picturing that scene from Rocky where Stallone eats all those raw eggs.
Most of the dietary cholesterol in Western communities is derived from eggs, but science has found no evidence which relates the number of eggs consumed to heart disease. A recent study from Rockefeller University gave participants either three servings (300 grams) of shrimps or two large eggs a day, each providing 580mg of cholesterol. Researchers found that both groups had an increase in both the good HDL cholesterol and the less desirable LDL cholesterol, which they interpreted to mean that neither diet significantly altered cardiovascular risk.
If you like eggs but don't want the and still don't want the extra cholesterol, use egg whites. Egg whites contain no cholesterol. However, doing the Rocky thing might not be such a good idea since the egg shell may contain salmonella. It's probably best to use either cooked egg whites, dried egg whites or pasteurized liquid egg whites.
BLANCO7
New member
- Awards
- 0
:sgrin: oh yeeeuhhYea I heard the same.... that everything is better raw..
HAHA I guess!I don't think I could live with myself throwing away perfectly good egg yolks
hey AA is real crucial to weight loss, 6 raw eggs spread out the day will do, take 400-600mg of niacin to help with cholestrol
Hi Andrew - what do you mean 'help' with cholestrol?hey AA is real crucial to weight loss, 6 raw eggs spread out the day will do, take 400-600mg of niacin to help with cholestrol
wat up Jack, after a while egg yolks can take a toll on the your lipids, so I suggest niacin to combat this scenario, regardless, I do not recommend one to consume raw eggs everyday for than three months, just enough to get the recomp effects, BELIEVE ME they will come.Hi Andrew - what do you mean 'help' with cholestrol?
How about eggs 3 times a week? Thats how often I have them.
BBR
Member
- Awards
- 0
I eat one whole egg and 5 egg whites every freaking day, I will not hear a bad word spoken about my poultry friends!
Seriously though, if you have a problem with cholestrol and eat 3 eggs a day then yes, it might cause issues. For a healthy person though, I seriously doubt you're going to have any ill effects from eating an egg a day or something.
Seriously though, if you have a problem with cholestrol and eat 3 eggs a day then yes, it might cause issues. For a healthy person though, I seriously doubt you're going to have any ill effects from eating an egg a day or something.
4 omega-3 eggs for me a day the whole eggs
it is a new addition to my diet and i am loving it, also found some organic sprouted grain no flour bread! 17g of carb with 5g fiber per slice....
4 eggs
2 slices bread
1 cup 1% milk
some no fat high protein cheese
best breakfast ever
49g protein
24g fat
48g carbs
it is a new addition to my diet and i am loving it, also found some organic sprouted grain no flour bread! 17g of carb with 5g fiber per slice....
4 eggs
2 slices bread
1 cup 1% milk
some no fat high protein cheese
best breakfast ever
49g protein
24g fat
48g carbs
AndreAnabolic
Member
- Awards
- 0
when i feel like getting real big! i smash 7 whole eggs a cup of oats a large glass of milk 2 tbsp of peanutbutter a pomegranite and a banana every mornin for breakfast....been doin the 7 whole eggs for years, nothins wrong with my numbers.
sometimes when im lazy tho i throw a cup of egg whites and 2 whole eggs in a blender with some whey and gulp it down...but not that i read that article about raw egg whites inibiting protein digestion, i dont know what im going to do with my life.
sometimes when im lazy tho i throw a cup of egg whites and 2 whole eggs in a blender with some whey and gulp it down...but not that i read that article about raw egg whites inibiting protein digestion, i dont know what im going to do with my life.
BBR
Member
- Awards
- 0
I think eggs are an awesome breakfast good. Eggs and oats go a long way towards a solid diet 
jmill05
New member
- Awards
- 0
:thumbsup: Definitely agreed!!! Eggs are such a low-cost protein and should be implemented in every diet. Stick to the whites if you're worried about cholesterol.
That's only a guide, considering i have no idea what your hereditary reaction is to cholesterol. If you havent had a problem yet, i would say keep them in your diet my man!!!
That's only a guide, considering i have no idea what your hereditary reaction is to cholesterol. If you havent had a problem yet, i would say keep them in your diet my man!!!
BBR
Member
- Awards
- 0
Sadly come people lack the common sense to simply stay away from something if they're uncomfortable with possible complications or if they have doubts.
Great info here. I just eat as many eggs as I can. Cooked,hardboiled,whatever. TP's whole egg protein powder is awesome.
I have been eating whole eggs for quite some time, and my cholesterol levels are normal. I usually get 3-4 whole eggs at least 5 days a week.
BBR
Member
- Awards
- 0
Go Team Egg!
Brother
Member
- Awards
- 0
Rally the egg crackers! Mount up your egg beaters and let the clucking begin!!
we call them 'Bum Nuts' around these parts :thumbsup:
Brother
Member
- Awards
- 0
Does anyone know the nutritional value of say...an ostrich egg over a chicken egg...ect. Just egg for thought?
BBR
Member
- Awards
- 0
Ahhhh .... bigger? 
In short:
...In conclusion, with the use of stable isotope techniques, we were able to determine the amounts of egg protein escaping digestion and absorption in the small intestine after ingestion of a physiologic load. Native egg protein is malabsorbed to an important extent. The assimilation of egg protein is facilitated by heat-pretreatment, but remains incomplete. The excellent correlation found between digestibility values and 13C-breath test data validated the 13C-egg protein breath test as an acurate alternative for the evaluation of protein digestibilty.....the true ileal digestibility of cooked and raw egg protein amounted to 90.0+/-0.8 and 51.3+/-9.8%, respectively....the true ileal digestibility of egg protein is significantly enhanced by heat-pretreatment...
.
wow dude re read ur own post it talks about digestibility
anytime u cook anything protines break down and are less BIO avalible
BIO yeas when i add heat BIO means life and heat kills life( in the case of eggs)
but it comes down to if u really wana grow eate eggs WHOLE eggs look at the tabe in the begening of the thread, the yoke holds most of the nutriants
by not eating the yokes ur cuting yer self short. and who likes coming up short?
The bio availability of the egg protein is increased when it is cooked from like, 50%-90%.anytime u cook anything protines break down and are less BIO avalible
if ur refuring to the studie postd above it is not talkin about bio availabilityThe bio availability of the egg protein is increased when it is cooked from like, 50%-90%.
i c wat ur sayin like soft boild eggs
natas9
New member
- Awards
- 0
wow dude re read ur own post it talks about digestibility
anytime u cook anything protines break down and are less BIO avalible
BIO yeas when i add heat BIO means life and heat kills life( in the case of eggs)
but it comes down to if u really wana grow eate eggs WHOLE eggs look at the tabe in the begening of the thread, the yoke holds most of the nutriants
by not eating the yokes ur cuting yer self short. and who likes coming up short?
Um...You can correct me on how I phrased it. That's fine. But maybe you don't get it; If it isn't being digested and absorbed well it isn't very bioavailable to you now is it?
You wanna eat raw eggs? Knock yerself out.
Btw, I'm well aware of the nutritional value of egg yolks.
gekkoboy14
New member
- Awards
- 0
misinformed doctors believe that saturated fats contribute to cholesterol.Eggs are high in cholesterol in fact one large egg has about 213 milligrams (mg) of cholesterol — all of which is found in the yolk. We also suspect that a diet high in cholesterol can contribute to elevated blood cholesterol levels. However, the extent to which dietary cholesterol raises blood cholesterol levels isn't clear. Many scientists believe that saturated fats and trans fats have a greater impact than does dietary cholesterol in raising blood cholesterol.
Most of the dietary cholesterol in Western communities is derived from eggs, but science has found no evidence which relates the number of eggs consumed to heart disease. A recent study from Rockefeller University gave participants either three servings (300 grams) of shrimps or two large eggs a day, each providing 580mg of cholesterol. Researchers found that both groups had an increase in both the good HDL cholesterol and the less desirable LDL cholesterol, which they interpreted to mean that neither diet significantly altered cardiovascular risk.
If you like eggs but don't want the and still don't want the extra cholesterol, use egg whites. Egg whites contain no cholesterol. However, doing the Rocky thing might not be such a good idea since the egg shell may contain salmonella. It's probably best to use either cooked egg whites, dried egg whites or pasteurized liquid egg whites.
u can eat 6 eggs aday raw and only increase ur chance of getting salmonella in yer whole life by 50%
just look up the risk of salmonella and eggs ther r countless studies
just look up the risk of salmonella and eggs ther r countless studies
BBR
Member
- Awards
- 0
I'd prefer to cook them either way!
| Thread starter | Similar threads | Forum | Replies | Date |
|---|---|---|---|---|
|
|
Eggs - Cheap vs. Expensive Types | Nutrition / Health | 21 | |
|
|
I love RAW EGGS | Nutrition / Health | 71 | |
|
|
Eggs whites post workout | Supplements | 68 | |
|
|
Unanswered Raw eggs? | Nutrition / Health | 15 | |
|
|
EGGS are bad for you - article | Nutrition / Health | 49 |