some info on thp ethers....
------------
Tetrahydropyranyl-ethers (THP-ethers) and other acetals
Chemists use THP ethers for temporary protection of hydroxyl groups against undesired reactions. Later the THP group can be removed easily by acid hydrolysis. Anabolic steroids are often marketed as THP ethers following the same idea. In the body the THP group is removed to set free the active steroid.
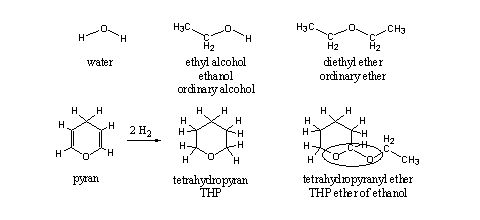
THP ethers of steroids are only partial ethers, although the word ether is in the name. An ether can be derived from water by substituting the H-atoms by alkyl groups. In Scheme 1 first one H-atom is replaced by an ethyl group, which leads to ethanol, the common alcohol in beer, wine and stronger alcoholic beverages. When also the second H-atom is replaced by an ethyl group, an ether is formed, in this case diethyl ether, the most common ether. In an ether both C-atoms next to the O-atom have only one bond to this O-atom.
A sixmembered ring with an O-atom and two double bonds is called a pyran. When both double bonds are reduced, each with two H-atoms, four H-atoms will enter the molecule and we get tetrahydropyran (see Scheme 1). When the second H-atom at the O-atom of ethanol is replaced by this group, we get the tetrahydropyranyl ether of ethanol. Till here everything is the same in the upper and lower row of Scheme 1. Nevertheless there are differences between the two ethers.
Scheme 1
On the right side of the O-atoms everything is the same, but on the left side this is not the case. In diethyl ether the left C-atom also has only one bond to the O-atom. In the tetrahydropyranyl ether this left C-atom has two bonds to two different O-atoms.
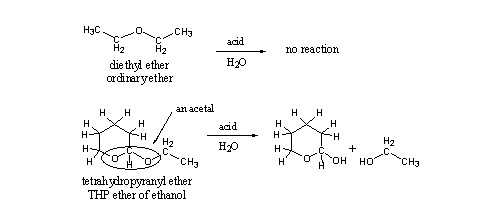
This seemingly small difference in structure means that this group has other chemical properties than an ordinary ether, and it has also a different name. This group is encircled in the structural formula and is called an acetal. Acetals are easily hydrolyzed under acidic conditions and ethers are not (see Scheme 2). The explanation for this difference in chemical behavior is known but we will not go into that in this book.
Scheme 2
The stomach contains gastric acid. Chemists know this acid better as hydrochloric acid (HCl) and this is a strong acid. Gastric acid causes a pH between 1 and 2 in the stomach and that are pretty strong acidic conditions. Chemists indicate acidity with pH values. Ordinary water has a pH of 7, which is called neutral. A pH between 0 and 7 is called acidic and a pH between 7 and 14 is called basic. In the intestinal tract the pH is slightly basic, between 7 and 8. In the blood and in muscles the pH is close to neutral.
Acetals like THP ethers hydrolyze easily under acidic conditions, which means that orally taken THP ethers of anabolic steroids will hydrolyze already in the stomach. This sets free the anabolic steroid, while it still has to pass the intestinal track and the liver. So there will be time and opportunity for metabolic transformation of the steroid to inactive metabolites.
A THP ether also can be given parentally, but the THP steroid derivative itself is not active. When the derivative does not pass the stomach, the THP ether group has to be removed in a different way. In the blood and in muscles the pH is neutral and under these conditions THP ethers are stable compounds. We also do not have enzymes in the blood, which can hydrolyze THP ethers, as is the case with esters.
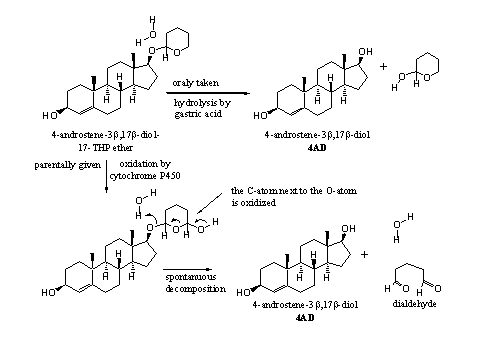
However, cytochrome P450 enzymes can oxidize THP ethers in the liver. This oxidation takes place in the ring next to the O-atom. The oxidation product then decomposes spontaneously to a dialdehyde and the free steroid (see Scheme 3). This scheme is a bit speculative, because little has been published about the metabolism of THP ethers.
Scheme 3
The free steroid will be converted in the liver to glucuronates or sulfate esters. The free steroid also may be inactivated through bonding to Sex Hormone Binding Globuline. Probably only a small part of the steroid will reach the muscle cell.
There is a clear difference between esters and THP-ethers of anabolic steroids. Esters are given parentally and after a slow release in the blood, enzymes will hydrolyze them to the active anabolic steroid. Orally taken THP ethers will hydrolyze already in the stomach. Parentally given THP ethers are oxidized in the liver and the active steroid is set free there.
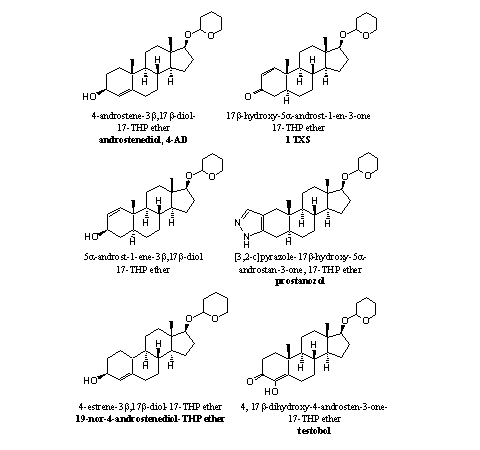
As orally taken THP ethers are already hydrolyzed in the stomach, it does not make much difference when you take the anabolic steroids itself or its THP ether. The marketing of an anabolic steroid as its THP ether probably has more to do with avoidance of patents or law regulations than with the making of better or longer lasting anabolic compounds. In Figure 1 some THP ethers of anabolic steroids are collected, which we have found in nutritional supplements.