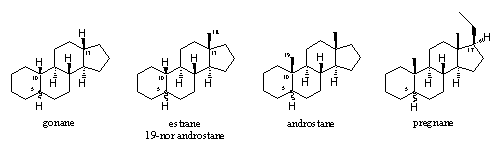
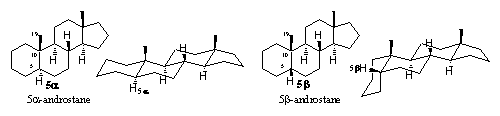Nowadays computer programs are available that convert chemical names into structural formulas and visa versa. A good example of such a program is the chemical drawing program called Chem draw.
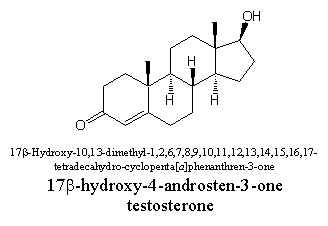
Chemists themselves have usually been among the first to create confusion in nomenclature, mostly for historical reasons. A compound like testosterone has been isolated for the first time from testes and is therefore called after that. Because the compound contains a steroid skeleton and a carbonyl group (a ketone group), the suffixes –ster and –one have been taken together and have been added. In this way the name testosterone is composed.
After some time more compounds with steroid skeletons are discovered and named in a similar way. When the number of compounds in the same group is large enough it becomes necessary to regulate their nomenclature internationally and after some or much discussion, rules are established. This is a time consuming process in which many authorities put forward their preferences. In the meantime several names have started to circulate, sometimes for the same compound.
Finally the IUPAC (Intenational Union of Pure and Applied Chemistry) takes a decision about the nomenclature for a class of compounds and after that these systematic names are used in the official chemical literature. However, the non official names are often firmly rooted and remain in use for years and years, especially in other sciences outside chemistry. Not seldom the official name is so complicated that nobody will use it anyhow and the historically established trivial name is maintained.
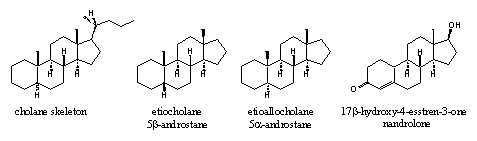
On the labels of bottles with pils and in publications about anabolic steroids, often the name etiocholane can be found. The prefix etio means degraded in Greek, so this name indicates a degraded cholane skeleton. This is an old-fashioned name for the 5b-androstane skeleton (see Figure 2), which originates from before the 2e world war.
The more common 5a-androstane skeleton is indicated in this nomenclature system with the name etioallocholane (see Figure 4). The Greek prefix allo indicates a small difference and is used also for other steroid skeletons with a 5a configuration.
Double bonds are mentioned as etiocholene and usually the double bond is located between C2 and C3. When the double bond is located at other positions, the prefix D is used in combination with the lowest number of the C-atoms on either side of the double bond.
After the 2e world war, an easier and straightforward nomenclature system has been adopted in 1950. This system is described above and generally used in the chemical literature [1]. Regularly updates of the nomenclature system have been published [2].
However in other sciences like biology, physiology, endocrinology, medicinal science and food science the old names are encountered often, although scientists have been urged regularly to use the correct steroid nomenclature [3].
In advertising and trade old fashioned names for anabolic steroids can be found every day. This confusing and misleading situation should come to an end as soon as possible. History learns however that this is not an easy task [3].
1. Steroid Nomenclature, Chemistry and Industry, London, (1950), 1-11; Steroid Nomenclature, Journal of the Chemical Society, (1951), 3526.
2. Nomenclature of steroids, Pure and Applied Chemistry, (1972), vol. 1, 2, 285.
3. D.E. Kime, Steroid Nomenclature, General and Comparative Endocrinology, (1995), 98, 119.
 It's a little older, but still appears to be relevant. It explains a few things like -ane vs -ene, methyl vs ethyl, gona(ane) vs estra(ane), etc.
It's a little older, but still appears to be relevant. It explains a few things like -ane vs -ene, methyl vs ethyl, gona(ane) vs estra(ane), etc.