Random987
New member
- Awards
- 0
I've been keeping one eye on the development of SARMS (Selective Androgen Receptor Modulator). It appears many of the labs are able to pin point exactly what receptors to bind to, which avoids many problems associated with regular TRT. So, you can design a SARM that has anabolic, libidio enhancing characteristics that will not effect hair loss (or may even grow hair), will not convert to estrogen, will not damage liver function, blood pressure or lipids. Also, it is ORALLY available.
here is a recent abstract:
Endocrinology. 2006 Oct 5; [Epub ahead of print] Links
An Orally-Active Selective Androgen Receptor Modulator is Efficacious on Bone, Muscle and Sex Function with Reduced Impact on Prostate.
* Miner JN,
* Chang W,
* Chapman MS,
* Finn PD,
* Hong MH,
* Lopez FJ,
* Marschke KB,
* Rosen J,
* Schrader W,
* Turner R,
* van Oeveren A,
* Viveros H,
* Zhi L,
* Negro-Vilar A.
Research and Development, Ligand Pharmaceuticals, 10275 Science Center Drive, San Diego, CA 92121; NIEHS/NIH - Environmental Diseases & Medicine Program, Research Triangle Park, NC; Metabasis Therapeutics, Inc. 11119 North Torrey Pines Rd., La Jolla, CA 92037; Department of Nutrition and Exercise Sciences, Oregon State University, 127 Milam Hall, Corvallis, OR 97331.
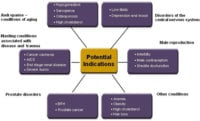
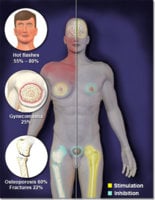
A number of conditions, including osteoporosis, frailty and sexual dysfunction in both men and women have been improved using androgens. However, androgens are not widely used for these indications because of the side effects associated with these drugs. We describe an androgen receptor ligand that maintains expected anabolic activities with substantially diminished activity in the prostate. LGD2226 is a non-steroidal, non-aromatizable, highly selective ligand for the androgen receptor (AR), exhibiting virtually no affinity for the other intracellular receptors. We determined that AR bound to LGD2226 exhibits a unique pattern of protein-protein interactions compared with testosterone, fluoxymesterone (an orally available steroidal androgen) and other steroids, suggesting that LGD2226 alters the conformation of the ligand binding domain (LBD). We demonstrated that LGD2226 is fully active in cell-based models of bone and muscle. LGD2226 exhibited anabolic activity on muscle and bone with reduced impact on prostate growth in rodent models. Biomechanical testing of bones from animals treated with LGD2226 showed strong enhancement of bone strength above sham levels. LGD2226 was also efficacious in a sex behavior model in male rats measuring mounts, intromissions, ejaculations and copulation efficiency. These results with an orally-available, non-aromatizable androgen demonstrate the important role of the androgen receptor and androgens in mediating a number of beneficial effects in bone, muscle and sexual function independent from the conversion of androgens into estrogenic ligands. Taken together, these results suggest that orally-active, non-steroidal SARMs may be useful therapeutics for enhancing muscle, bone and sexual function.
PMID: 17023534 [PubMed - as supplied by publisher]
here is a recent abstract:
Endocrinology. 2006 Oct 5; [Epub ahead of print] Links
An Orally-Active Selective Androgen Receptor Modulator is Efficacious on Bone, Muscle and Sex Function with Reduced Impact on Prostate.
* Miner JN,
* Chang W,
* Chapman MS,
* Finn PD,
* Hong MH,
* Lopez FJ,
* Marschke KB,
* Rosen J,
* Schrader W,
* Turner R,
* van Oeveren A,
* Viveros H,
* Zhi L,
* Negro-Vilar A.
Research and Development, Ligand Pharmaceuticals, 10275 Science Center Drive, San Diego, CA 92121; NIEHS/NIH - Environmental Diseases & Medicine Program, Research Triangle Park, NC; Metabasis Therapeutics, Inc. 11119 North Torrey Pines Rd., La Jolla, CA 92037; Department of Nutrition and Exercise Sciences, Oregon State University, 127 Milam Hall, Corvallis, OR 97331.
A number of conditions, including osteoporosis, frailty and sexual dysfunction in both men and women have been improved using androgens. However, androgens are not widely used for these indications because of the side effects associated with these drugs. We describe an androgen receptor ligand that maintains expected anabolic activities with substantially diminished activity in the prostate. LGD2226 is a non-steroidal, non-aromatizable, highly selective ligand for the androgen receptor (AR), exhibiting virtually no affinity for the other intracellular receptors. We determined that AR bound to LGD2226 exhibits a unique pattern of protein-protein interactions compared with testosterone, fluoxymesterone (an orally available steroidal androgen) and other steroids, suggesting that LGD2226 alters the conformation of the ligand binding domain (LBD). We demonstrated that LGD2226 is fully active in cell-based models of bone and muscle. LGD2226 exhibited anabolic activity on muscle and bone with reduced impact on prostate growth in rodent models. Biomechanical testing of bones from animals treated with LGD2226 showed strong enhancement of bone strength above sham levels. LGD2226 was also efficacious in a sex behavior model in male rats measuring mounts, intromissions, ejaculations and copulation efficiency. These results with an orally-available, non-aromatizable androgen demonstrate the important role of the androgen receptor and androgens in mediating a number of beneficial effects in bone, muscle and sexual function independent from the conversion of androgens into estrogenic ligands. Taken together, these results suggest that orally-active, non-steroidal SARMs may be useful therapeutics for enhancing muscle, bone and sexual function.
PMID: 17023534 [PubMed - as supplied by publisher]