When is this coming out? I'm stoked beyond belief to have a DHT based product on the market. Sounds like a replacement for m5aa :head:
i believe ETA is 3-4 weeks...
You guys are awesome! I can't wait to combine RPM with The One! I honestly never expected something like this to be released from you guys. Cool.:thumbsup:i believe ETA is 3-4 weeks...
A week ago the ETA was 4-5 weeks. All the ingredients have now been released, so Austin is right in saying not long to go nowWhen is this coming out? I'm stoked beyond belief to have a DHT based product on the market. Sounds like a replacement for m5aa :head:
can we get the ingredients ? or we have to wait :trout:A week ago the ETA was 4-5 weeks. All the ingredients have now been released, so Austin is right in saying not long to go now
posted in the supplements as well as company promotions sectionscan we get the ingredients ? or we have to wait :trout:
so.....Will AN ship to me in Australia? I'll run the risk. It seems worth it. I been training so long, this will put the icing on the cake for me. Customs cant open EVERYTHING. Hopefully I can get it through, with a wee bit of luck. Will only need the 1 bottle if you get a good month out of it, cause I am only after a few kilo. Gotta get it, gotta get it......:think:
ozzy you could possibly get it through if you order through appnut direct
*Mr burns voice* exxxxcellent Smithers:thumbsup:ozzy you could possibly get it through if you order through appnut direct
Have you guys had any testers run this yet? I can imagine its gonna be harsh on the hairline.
yes we had 2 in the AN crew plus some others. None reported excessive hair loss and a some were prone to MPB
If you need testers, look my way!yes we had 2 in the AN crew plus some others. None reported excessive hair loss and a some were prone to MPB
The label will be released soon. In the meantime, as Austin said, ingredients are in the Technical Write-Up:can we get the ingredients ? or we have to wait :trout:
Name: 17a-methyl-etioallocholan-17b-ol-3-hydroxyimine
Standard: Testosterone
Androgenic: 140
Anabolic: 380
Estrogenic Activity: none
Progestational Activity: none
Derivation/Analog of: Dihydrotestosterone
It has been mentioned to you before that your best chance is to order from AN direct, or from somone who WILL send to Australia (i.e. Nutra Planet).so.....Will AN ship to me in Australia? I'll run the risk. It seems worth it. I been training so long, this will put the icing on the cake for me. Customs cant open EVERYTHING. Hopefully I can get it through, with a wee bit of luck. Will only need the 1 bottle if you get a good month out of it, cause I am only after a few kilo. Gotta get it, gotta get it......:think:
We have had a professional testing group, as well as two associated with AN test The ONE (test results will be made available when ALL results are in). In the meantime, both Eric and Joe have mentioned their experiences with The ONE:Have you guys had any testers run this yet? I can imagine its gonna be harsh on the hairline.
...i have never taken anything that left me feeling so good. high energy, strong focus, high libido, positive mood enhancement, the works. each morning i'd wake up impatient waiting for that first dose to kick in.
...
Ok so more of my results i gained
14lbs
around 1/4 inch on waist
7/8 inch on chest
3/8 on bis
between 1/8-1/4 on forearms
3/4 on shoulders
and tendonitis from pushing myself with the strength gains, which is why i stopped the cycle. I thought it was a sprain at first, but its not. so no lifting for me now for a while. suckage. I pushed myself hard because it felt so good, and obviously pushed too hard.
...
my lifting style isn't well suited to showing it, as i mostly use dumbbells and dont particularly lift for strenght gains. but I was up a good 10% on weights on pretty much everything, basically broke my all time prs on everything except squats (and I hadnt done legs at all for almost a month prior to starting this)
probably the exercise I noticed it the most on were lateral + front raises oddly enough. those 2 are ones I hadn't seen any weight progress on in a long time.
...
Blood Presssure: i monitored daily, have a machine at home. My normal is around 125/75, the absolute peak I measured on cycle about 2 hours after a workout was 134/82, most of the time was close enough to normal that I couldn't see a difference at all.
so the first week of NOV im up at appnut land and dirk starts showing me all this info and ideas. He shows me the write up the discovery all the good stuff.
then he says you wanna try it out and see what we get. I said sure why not. At this point we have no idea what to expect we have a few clues but this was a shot in the dark well after a week on it i started noticing so much good stuff. size weight, awesome feelings of well being, I was like wtf. Im too soon to jump ill chalk it up to placebo until i spoke with E on this who was getting similar results. we started talking to appnut about it. They were getting sim results with others who were trying. then it happened 8 days after first dose. Sat morning im cutting drywall and oops
...
so now im done. No gym no nothing. 2 weeks i didnt move increase in blood pressure caused more bleeding. finally 3 weeks later i started mild cardio and some leg work. it would be a full 6 weeks before i attempted uppperbody. but the whole time i kept dosing as laid out and I kept gaining size.
My first day back in the gym doing bench press with a full wrap and shield on my thumb. still bleeding i repped 4 sets of 285 for 8 reps. something i couldnt do before with 2 good hands. I called Dirk and was like wtf I havent touched upper body in 6 weeks and im much stronger then before
granted the rest was nice but man im now up 12 lbs total and i only lifted legs for 2 weeks
so yes I gained size while laying in bed out of work unable to do anything.
...
here is the best part its been 1 month since i reported my results and nothing has changed. I still have all my strength. I kept taking this because i just knew no activity for a hard gainer like myself would just shed the size off of me and yeah i was depressed laying in hospital as they were coterizing my thumb shut. i just knew i was gonna lose alot of gains made over the last yr in korea
i cant wait the next time i try this to actually be able to lift on this the feelings of well being were unlike anything ive ever tried i just felt fantastic
...
yes i have a receding hairline
and yes if ur going bald nothing u can do with stop this
5 weeks on this compound during initial trials didnt change anything in my
bp hairline, prostate, nothing of that nature.
We don't need more testers (since it is almost done). But there WILL be logging opportunities coming...If you need testers, look my way!
I am posting the write-up in this thread.
Guys, questions are welcome, but please read these two threads FIRST (as chances are, the answer is in there)!:
* http://anabolicminds.com/forum/company-promotions/112713-2009-s-biggest.html
* http://anabolicminds.com/forum/supplements/113105-here-s-write.html
The ONE: Part I - Introduction
OK, without further ado....
[size=+3]The ONE™[/size] by


Name: 17a-methyl-etioallocholan-17b-ol-3-hydroxyimine
Standard: Testosterone
Androgenic: 140
Anabolic: 380
Estrogenic Activity: none
Progestational Activity: none
Derivation/Analog of: Dihydrotestosterone
Brief History:
• Dihydrotestosterone (DHT) was first synthesized in 1935.
• Primary active ingredient (17a-methyl-etioallocholan-17b-ol-3-hydroxyimine) was first described by Z. Cekan and B. Pelc, Effects of 1-alkyl substitution on the biological action in a series of androstanes. Steroids 8,209 (1966).
• Never commercially produced – A COMPLETELY NOVEL MOLECULE.
Product Characteristics:
• The One is an oral prohormone of Dihydrotestosterone (DHT), a compound that produces effects 3 to 5 times more potent than testosterone on the androgen receptor.
• The One can inhibit the action of estrogens by acting as a competitive antagonist and/or decreasing the mRNA-induced transcription of estrogenic effects post-binding.
• The One can also act like an aromatase inhibitor, disallowing the conversion of androgens to estrogen.
• The One activates GLUT-4 receptors in skeletal muscle, increasing glycogen uptake of 17-alpha alkylated molecules for greater oral bioavailability.
• The One can significantly lower SHBG levels, allowing for more active product to reach skeletal muscle.
Effects Related to Physique Enhancement:
• Rapid lean body mass gains - typical users report 0.75 lb./day gains for first 2-3 weeks
• Overall hardening effect to physique
• Increased vascularity
• Increased strength
• Dramatically enhanced neural function (coordination) as DHT is the predominant androgen in neural function
• Mood-enhancing effect
• Lipolytic effect (increases fat metabolism)
ADMINISTRATION/DOSAGE:
• Effective dose: 67.5 - 90 mg/day (3-4 capsules) ; DO NOT EXCEED 90 MG/DAY
• Can be taken with or without food; preferably in the AM
• Oral Administration Only
• 4-5 week administration period; do not exceed 5 weeks duration
OK, without further ado....
[size=+3]The ONE™[/size] by


Name: 17a-methyl-etioallocholan-17b-ol-3-hydroxyimine
Standard: Testosterone
Androgenic: 140
Anabolic: 380
Estrogenic Activity: none
Progestational Activity: none
Derivation/Analog of: Dihydrotestosterone
Brief History:
• Dihydrotestosterone (DHT) was first synthesized in 1935.
• Primary active ingredient (17a-methyl-etioallocholan-17b-ol-3-hydroxyimine) was first described by Z. Cekan and B. Pelc, Effects of 1-alkyl substitution on the biological action in a series of androstanes. Steroids 8,209 (1966).
• Never commercially produced – A COMPLETELY NOVEL MOLECULE.
Product Characteristics:
• The One is an oral prohormone of Dihydrotestosterone (DHT), a compound that produces effects 3 to 5 times more potent than testosterone on the androgen receptor.
• The One can inhibit the action of estrogens by acting as a competitive antagonist and/or decreasing the mRNA-induced transcription of estrogenic effects post-binding.
• The One can also act like an aromatase inhibitor, disallowing the conversion of androgens to estrogen.
• The One activates GLUT-4 receptors in skeletal muscle, increasing glycogen uptake of 17-alpha alkylated molecules for greater oral bioavailability.
• The One can significantly lower SHBG levels, allowing for more active product to reach skeletal muscle.
Effects Related to Physique Enhancement:
• Rapid lean body mass gains - typical users report 0.75 lb./day gains for first 2-3 weeks
• Overall hardening effect to physique
• Increased vascularity
• Increased strength
• Dramatically enhanced neural function (coordination) as DHT is the predominant androgen in neural function
• Mood-enhancing effect
• Lipolytic effect (increases fat metabolism)
ADMINISTRATION/DOSAGE:
• Effective dose: 67.5 - 90 mg/day (3-4 capsules) ; DO NOT EXCEED 90 MG/DAY
• Can be taken with or without food; preferably in the AM
• Oral Administration Only
• 4-5 week administration period; do not exceed 5 weeks duration
The ONE: Part II - Test Data
Test Data:
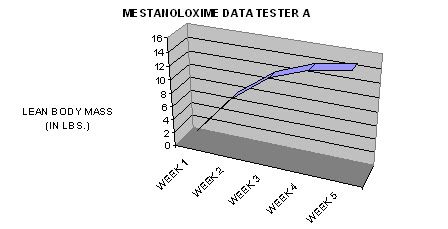
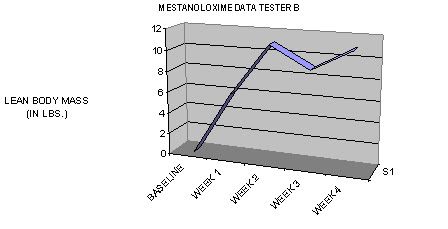
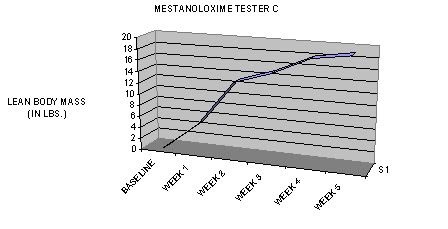
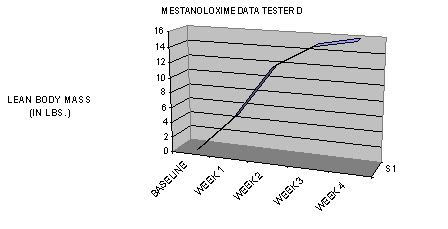
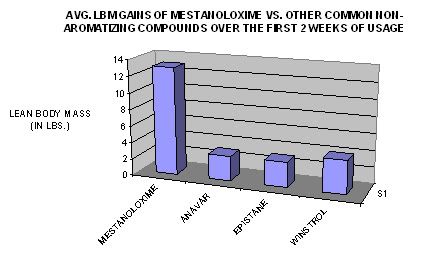
TIME-DEPENDANT COMPARISON TO OTHER NON-AROMATIZING ORAL PRODUCTS IN TERMS OF LEAN BODY MASS GAINS DURING THE FIRST TWO WEEKS OF USAGE:
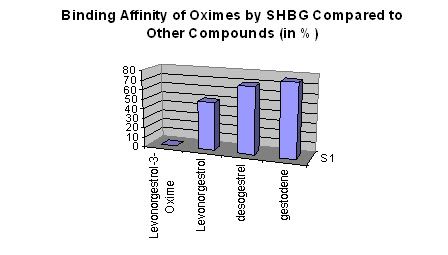
Binding of Oximes by Steroid Hormone Globulin (SHBG) Compared to Other Compounds:
Test Data:




TIME-DEPENDANT COMPARISON TO OTHER NON-AROMATIZING ORAL PRODUCTS IN TERMS OF LEAN BODY MASS GAINS DURING THE FIRST TWO WEEKS OF USAGE:

Binding of Oximes by Steroid Hormone Globulin (SHBG) Compared to Other Compounds:

The ONE: Part III - ON Cycle and PCT Tips
ON CYCLE: TIPS TO MAXIMIZE GROWTH
• Take Complete Balance™ Multi-Vitamin/Multi-Mineral
o Ensures that all essential vitamins and minerals are being taken in daily
• Take Omega Essentials™ EFA/DHA at 3-5 grams per day
o Strengthens cell membranes
o Enhances insulin sensitivity
o Synergistic with the GLUT-4 translocating ability of The One
o Enhances healthy HDL/LDL profiles
• Take Bio-Mend™ Anti-Oxidant formula
o High ORAC Value
o Protects cellular membrane
o Protects transcriptional factors (mRNA and DNA)
• Milk Thistle
o Hepatoprotectant – protects liver health
• In general, maintain a healthy diet and lifestyle:
o Drink Plenty of water- at least 64 oz. per day
o Ingest at least 1 gram of protein per lb. of body weight daily
o Sleep at least 7 hours per night
o Eat lots of fruits and vegetables
o Eat lots of complex carbs
o Eat 5-6 smaller protein and carb-rich meals throughout the day
o Increase calories to at least 500 Kcal/day over your normal intake
o BCAAs and Creatine will be helpful
o Avoid alcohol and tobacco
TRAINING
• 8-15 rep sets- 75-85% of 1RM Max for maximal hypertrophy
• Train 3-5 days per week
• Use Heavy Compound Exercises and Variations
• Squats
• Deadlifts
• Pull-Ups w/ multiple grips
• Bench Press
POST CYCLE: TIPS TO MAXIMIZE RECOVERY & MAINTAIN GAINS
6-BROMODIONE, 50 mg/day
• Aromatase Inhibitor
• Increases free testosterone with minimal endocrine suppression
BIO-MEND™ and COMPLETE BALANCE™ as directed on labels
• Prevent calcium leakage from Sarcoplasmic Reticulum
• Helps to maintain cell membrane integrity
• Helps to maintain healthy HDL/LDL levels
• Helps to protect mRNA and DNA
• Helps to keep adequate nutrient profile
OMEGA ESSENTIALS™ as directed on label
• Increases cell membrane strength
• Helps establish/maintain healthy lipid profiles
• Increases insulin sensitivity
DRIVE™ as directed on label
• Increases Cyclic AMP (cAMP) which in turn can have stimulatory effects on spermatogenesis
• Increases free testosterone
• Increases thyroid output
• Increases calcium uptake and decreases calcium leakage from sarcoplasmic reticulum (SR) increased calcium storage in SR helps to mediate a non-hormonal anabolic response
IGF-2® as directed on label
• Increases free testosterone
• Increases GH output
ON CYCLE: TIPS TO MAXIMIZE GROWTH
• Take Complete Balance™ Multi-Vitamin/Multi-Mineral
o Ensures that all essential vitamins and minerals are being taken in daily
• Take Omega Essentials™ EFA/DHA at 3-5 grams per day
o Strengthens cell membranes
o Enhances insulin sensitivity
o Synergistic with the GLUT-4 translocating ability of The One
o Enhances healthy HDL/LDL profiles
• Take Bio-Mend™ Anti-Oxidant formula
o High ORAC Value
o Protects cellular membrane
o Protects transcriptional factors (mRNA and DNA)
• Milk Thistle
o Hepatoprotectant – protects liver health
• In general, maintain a healthy diet and lifestyle:
o Drink Plenty of water- at least 64 oz. per day
o Ingest at least 1 gram of protein per lb. of body weight daily
o Sleep at least 7 hours per night
o Eat lots of fruits and vegetables
o Eat lots of complex carbs
o Eat 5-6 smaller protein and carb-rich meals throughout the day
o Increase calories to at least 500 Kcal/day over your normal intake
o BCAAs and Creatine will be helpful
o Avoid alcohol and tobacco
TRAINING
• 8-15 rep sets- 75-85% of 1RM Max for maximal hypertrophy
• Train 3-5 days per week
• Use Heavy Compound Exercises and Variations
• Squats
• Deadlifts
• Pull-Ups w/ multiple grips
• Bench Press
POST CYCLE: TIPS TO MAXIMIZE RECOVERY & MAINTAIN GAINS
6-BROMODIONE, 50 mg/day
• Aromatase Inhibitor
• Increases free testosterone with minimal endocrine suppression
BIO-MEND™ and COMPLETE BALANCE™ as directed on labels
• Prevent calcium leakage from Sarcoplasmic Reticulum
• Helps to maintain cell membrane integrity
• Helps to maintain healthy HDL/LDL levels
• Helps to protect mRNA and DNA
• Helps to keep adequate nutrient profile
OMEGA ESSENTIALS™ as directed on label
• Increases cell membrane strength
• Helps establish/maintain healthy lipid profiles
• Increases insulin sensitivity
DRIVE™ as directed on label
• Increases Cyclic AMP (cAMP) which in turn can have stimulatory effects on spermatogenesis
• Increases free testosterone
• Increases thyroid output
• Increases calcium uptake and decreases calcium leakage from sarcoplasmic reticulum (SR) increased calcium storage in SR helps to mediate a non-hormonal anabolic response
IGF-2® as directed on label
• Increases free testosterone
• Increases GH output
The ONE: Part IV - Technical Write-Up
Mechanism of Action/Function:
Absorption
• User ingests compound orally
• Digestion occurs in roughly 20-30 minutes (as with most encapsulated products)
• Compound is absorbed by the duodenum and jejunum of the small intestine, and then is displaced into the hepatic portal vein, (a special blood vessel leading from the digestive tract to the liver) where the compound must undergo the “first pass” through the liver and survive hepatic metabolism.
• Methylation allows the compound to survive the first pass through the liver, protecting the molecule from hepatic enzymatic degradation.
• After first pass, the compound is distributed into the inferior vena cava, and then to the heart for systemic distribution.
Distribution
• The heart pumps the active compound into all different areas of the body during the distribution phase. As is the case with most compounds, SHBG (steroid hormone binding globulin) can remove active compounds from systemic distribution before they reach their target organ (in this case, skeletal muscle).
• Binding proteins such as SHBG are rendered fairly inactive in this situation with the active ingredient in The One, because it is one of only a few compounds that does not carry an actual 3-keto group; it carries a 3-oxime group instead.
• Research has dictated that oximes have little or no binding affinity to SHBG in the blood stream, allowing for more free active substrate to reach their target (the androgen receptor (AR) in skeletal muscle). In essence, the 3-oxime “protects” the molecule from being bound (see graph below) leaving more active substrate in the blood to target skeletal muscle.
Metabolism
• Once The One reaches skeletal muscle, it must interact with specific target androgen receptors to exert the desired effect of increased protein synthesis and increased nitrogen retention.
• Under normal circumstances, DHT is quickly deactivated in the blood by 3a-HSD into 5alpha-androstan-3a,17b-diol, a weak androgen, and its access to target skeletal muscle tissue is restricted.
• There is some evidence that the 3-oxime substituted for 3-keto group may slow this deactivation, allowing for greater binding of the molecule to the AR.
• Oximes are a group of compounds with the general formula R¹R²CNOH, where R1 constitutes an organic side chain, and R2 is another organic group. This forms a ketoxime.
• Oximes exist as two geometric stereoisomers: a syn-isomer and an anti-isomer.
• Oximes are formed by the action of hydroxylamine on ketones.
Receptor Binding
• Because the evidence suggests that active DHT may not be metabolized by 3a-HSD into the weaker 5-alpha-androstan-3a,17b-diol within the skeletal muscle because of the oxime, there is a very good chance that fairly large amounts of DHT will actually bind to the AR in skeletal muscle.
• DHT has a 3 to 5 times greater binding potency on the androgen receptor than testosterone, so the resulting effect from The One is a very powerful growth stimulus on skeletal muscle.
• Androgens can act in a genomic (transcriptive, from direct AR binding) or non-genomic (non-AR binding) fashion on target cells.
• Some examples of non-genomic effects: increased IGF-1 expression, displacement of estrogen and progesterone from receptor sites, displacement of aromatase, and increased uptake of calcium in the sarcoplasmic reticulum.
Genomic Effects and MOA:
• The AR has four functional regions: a hinge region containing a nuclear localization signal, a carboxy-terminal ligand-binding domain (AF-2 site), an amino terminal regulatory domain (AF-1 site) and a DNA-binding domain composed of two zinc fingers.
• ARs that are not ligated are located primarily in the cytoplasm, and are bound to heat shock proteins (HSPs). HSPs stabilize the tertiary structure of the AR, permitting androgen binding.
• When The One binds the AR, there is a resulting dissociation of HSPs from the AR, allowing for dimerization (to be held together by molecular force) of the AR and subsequent tyrosine kinase phosphorylation, resulting in translocation (movement) of the AR to the nucleus.
• When the AR translocates inside the nucleus, it binds androgen response elements located in the enhancer and promoter areas of target genes. This results in the consequential assembly of regulatory proteins, and the creation of an active complex for transcription.
• The regulatory proteins form a cross-bridge with the AR, the RNA polymerase, and the pre-initiation complex. Specific coactivators begin transcription by recruiting protein clusters to DNA that change the chromatin scaffolding to a form that is more active for transcription.
• The resulting activated transcription results in synthesis of mRNA, which is signaled by ribosomes to produce specific proteins. Changes in specific cell proteins follow, which in turn mediate growth responses, nitrogen balance, and protein synthesis, among other things.
Excretion
• The Compound is removed from the body via excretion, through the kidneys via glomelular filtration.
WARNING: This product is only intended to be consumed by healthy adult males 21 years of age or older. Not for use by women. Before using this product, consult with your physician if you are using any prescription or over the counter medicine or if you are unaware of your current medical condition. Do not use this product if you have any pre-existing medical condition including but not limited to: high or low blood pressure, cardiac arrhythmia, high cholesterol, stroke, heart, liver, kidney or thyroid disease, seizure disorder, psychiatric disease, diabetes, difficulty urinating due to prostate enlargement or if you are taking and MAO-B inhibitor or any other medication. Discontinue use and consult your health care professional if you experience any adverse reaction to this product. Possible androgenic side effects including but not limited to acne, increased risk of male pattern baldness, testicular atrophy and gynecomastia (males), may occur. Do not consume alcohol while taking this product. Do not exceed recommended serving. Store in a cool, dry place with lid tightly closed. Do not use if inner safety seal is broken or missing. KEEP OUT OF REACH OF CHILDREN.
Note: This product may contain ingredients which are banned by some athletic or government associations (including military).
Mechanism of Action/Function:
Absorption
• User ingests compound orally
• Digestion occurs in roughly 20-30 minutes (as with most encapsulated products)
• Compound is absorbed by the duodenum and jejunum of the small intestine, and then is displaced into the hepatic portal vein, (a special blood vessel leading from the digestive tract to the liver) where the compound must undergo the “first pass” through the liver and survive hepatic metabolism.
• Methylation allows the compound to survive the first pass through the liver, protecting the molecule from hepatic enzymatic degradation.
• After first pass, the compound is distributed into the inferior vena cava, and then to the heart for systemic distribution.
Distribution
• The heart pumps the active compound into all different areas of the body during the distribution phase. As is the case with most compounds, SHBG (steroid hormone binding globulin) can remove active compounds from systemic distribution before they reach their target organ (in this case, skeletal muscle).
• Binding proteins such as SHBG are rendered fairly inactive in this situation with the active ingredient in The One, because it is one of only a few compounds that does not carry an actual 3-keto group; it carries a 3-oxime group instead.
• Research has dictated that oximes have little or no binding affinity to SHBG in the blood stream, allowing for more free active substrate to reach their target (the androgen receptor (AR) in skeletal muscle). In essence, the 3-oxime “protects” the molecule from being bound (see graph below) leaving more active substrate in the blood to target skeletal muscle.
Metabolism
• Once The One reaches skeletal muscle, it must interact with specific target androgen receptors to exert the desired effect of increased protein synthesis and increased nitrogen retention.
• Under normal circumstances, DHT is quickly deactivated in the blood by 3a-HSD into 5alpha-androstan-3a,17b-diol, a weak androgen, and its access to target skeletal muscle tissue is restricted.
• There is some evidence that the 3-oxime substituted for 3-keto group may slow this deactivation, allowing for greater binding of the molecule to the AR.
• Oximes are a group of compounds with the general formula R¹R²CNOH, where R1 constitutes an organic side chain, and R2 is another organic group. This forms a ketoxime.
• Oximes exist as two geometric stereoisomers: a syn-isomer and an anti-isomer.
• Oximes are formed by the action of hydroxylamine on ketones.
Receptor Binding
• Because the evidence suggests that active DHT may not be metabolized by 3a-HSD into the weaker 5-alpha-androstan-3a,17b-diol within the skeletal muscle because of the oxime, there is a very good chance that fairly large amounts of DHT will actually bind to the AR in skeletal muscle.
• DHT has a 3 to 5 times greater binding potency on the androgen receptor than testosterone, so the resulting effect from The One is a very powerful growth stimulus on skeletal muscle.
• Androgens can act in a genomic (transcriptive, from direct AR binding) or non-genomic (non-AR binding) fashion on target cells.
• Some examples of non-genomic effects: increased IGF-1 expression, displacement of estrogen and progesterone from receptor sites, displacement of aromatase, and increased uptake of calcium in the sarcoplasmic reticulum.
Genomic Effects and MOA:
• The AR has four functional regions: a hinge region containing a nuclear localization signal, a carboxy-terminal ligand-binding domain (AF-2 site), an amino terminal regulatory domain (AF-1 site) and a DNA-binding domain composed of two zinc fingers.
• ARs that are not ligated are located primarily in the cytoplasm, and are bound to heat shock proteins (HSPs). HSPs stabilize the tertiary structure of the AR, permitting androgen binding.
• When The One binds the AR, there is a resulting dissociation of HSPs from the AR, allowing for dimerization (to be held together by molecular force) of the AR and subsequent tyrosine kinase phosphorylation, resulting in translocation (movement) of the AR to the nucleus.
• When the AR translocates inside the nucleus, it binds androgen response elements located in the enhancer and promoter areas of target genes. This results in the consequential assembly of regulatory proteins, and the creation of an active complex for transcription.
• The regulatory proteins form a cross-bridge with the AR, the RNA polymerase, and the pre-initiation complex. Specific coactivators begin transcription by recruiting protein clusters to DNA that change the chromatin scaffolding to a form that is more active for transcription.
• The resulting activated transcription results in synthesis of mRNA, which is signaled by ribosomes to produce specific proteins. Changes in specific cell proteins follow, which in turn mediate growth responses, nitrogen balance, and protein synthesis, among other things.
Excretion
• The Compound is removed from the body via excretion, through the kidneys via glomelular filtration.
WARNING: This product is only intended to be consumed by healthy adult males 21 years of age or older. Not for use by women. Before using this product, consult with your physician if you are using any prescription or over the counter medicine or if you are unaware of your current medical condition. Do not use this product if you have any pre-existing medical condition including but not limited to: high or low blood pressure, cardiac arrhythmia, high cholesterol, stroke, heart, liver, kidney or thyroid disease, seizure disorder, psychiatric disease, diabetes, difficulty urinating due to prostate enlargement or if you are taking and MAO-B inhibitor or any other medication. Discontinue use and consult your health care professional if you experience any adverse reaction to this product. Possible androgenic side effects including but not limited to acne, increased risk of male pattern baldness, testicular atrophy and gynecomastia (males), may occur. Do not consume alcohol while taking this product. Do not exceed recommended serving. Store in a cool, dry place with lid tightly closed. Do not use if inner safety seal is broken or missing. KEEP OUT OF REACH OF CHILDREN.
Note: This product may contain ingredients which are banned by some athletic or government associations (including military).
The ONE: Part V - Studies and Clinical Information
STUDIES/CLINICAL INFORMATION:
• Kuhnz W, Fritzemeier KH, Hegele-Hartung C, Krattenmacher R. Comparative progestational activity of norgestimate, levonorgestrel-oxime and levonorgestrel in the rat and binding of these compounds to the progesterone receptor. Contraception. 1995 Feb;51(2):131-9.
• Li QG, Li DZ. Analysis of anti-implantational action of norethisterone-3-oxime. Contraception. 1987 Dec;36(6):667-76.
• Li DZ, Fu GM, Zuo MD, Zhou MY, Zhao XL, Wang GP, Wei ZN. Pharmacokinetic studies of norethisterone-3-oxime and norethisterone-3-oxime acetate in rhesus monkey. J Tongji Med Univ. 1991;11(2):81-7.
• Li QG. Aromatization and hydrolysis of norethisterone-3-oxime in rabbit. J Steroid Biochem Mol Biol. 1994 Jun;49(2-3):227-31.
• Hammerstein J. Prodrugs: advantage or disadvantage? Am J Obstet Gynecol. 1990 Dec;163(6 Pt 2):2198-203.
• Shrivastav TG, Basu A, Kariya KP. One step enzyme linked immunosorbent assay for direct estimation of serum testosterone. J Immunoassay Immunochem. 2003;24(2):205-17.
• Tripathi V, Nara S, Chaube SK, Rangari K, Saroha A, Kariya KP, Singh H, Shrivastav TG. J Development of rapid and sensitive one-step direct enzyme linked immunosorbent assay for 17-alpha-OH-progesterone in serum. Immunoassay Immunochem. 2008; 29(2):117-27.
• Nara S, Tripathi V, Chaube SK, Rangari K, Singh H, Kariya KP, Shrivastav TG. A novel enzyme-linked immunosorbent assay for cortisol using a long-chain biotinylated cortisol-3-CMO derivative. J Immunoassay Immunochem. 2008;29(4):390-405.
• Sinnecker G, Köhler S Sex hormone-binding globulin response to the anabolic steroid stanozolol: evidence for its suitability as a biological androgen sensitivity test. J Clin Endocrinol Metab. 1989 Jun;68(6):1195-200.
• Saartok T, Dahlberg E, Gustafsson JA. Relative binding affinity of anabolic-androgenic steroids: comparison of the binding to the androgen receptors in skeletal muscle and in prostate, as well as to sex hormone-binding globulin. Endocrinology. 1984 Jun;114(6):2100-6.
• Tóth M, Zakár T. Relative binding affinities of testosterone, 19-nortestosterone and their 5 alpha-reduced derivatives to the androgen receptor and to other androgen-binding proteins: a suggested role of 5 alpha-reductive steroid metabolism in the dissociation of "myotropic" and "androgenic" activities of 19-nortestosterone. J Steroid Biochem. 1982 Dec;17(6):653-60.
• Lu NZ, Wardell SE, Burnstein KL, Defranco D, Fuller PJ, Giguere V, Hochberg RB, McKay L, Renoir JM, Weigel NL, Wilson EM, McDonnell DP, Cidlowski JA (December 2006). "International Union of Pharmacology. LXV. The pharmacology and classification of the nuclear receptor superfamily: glucocorticoid, mineralocorticoid, progesterone, and androgen receptors". Pharmacol. Rev. 58 (4): 782–97.
• Hammond GL, Abrams LS, Creasy GW, Natarajan J, Allen JG, Siiteri PK.
• Serum distribution of the major metabolites of norgestimate in relation to its
• pharmacological properties. Contraception. 2003 Feb;67(2):93-9.
• Juchem M, Pollow K, Elger W, Hoffmann G, Möbus V. Receptor binding of norgestimate--a new orally active synthetic progestational compound. Contraception. 1993 Mar;47(3):283-94.
• Ouyang G, Chen Z, Cai XJ, Song BA, Bhadury PS, Yang S, Jin LH, Xue W, Hu DY, Zeng S. Synthesis and antiviral activity of novel pyrazole derivatives containing oxime esters group. Bioorg Med Chem. 2008 Nov 15;16(22):9699-707. Epub 2008 Oct 2.
• Shrivastav TG, Kanaujia PK. Direct radioimmunoassay for the measurement of serum testosterone using 3H as label. J Immunoassay Immunochem. 2007;28(2):127-36.
• Basu A, Shrivastav TG One step enzyme linked immunosorbent assay for direct estimation of serum cortisol. J Immunoassay. 2000 Feb;21(1):39-50.
• Koji Sato, Motoyuki Iemitsu, Katsuji Aizawa, and Ryuichi Ajisaka Testosterone and DHEA activate the glucose metabolism-related signaling pathway in skeletal muscle. Am J Physiol Endocrinol Metab 294: E961-E968, 2008.
• Davison SL, Bell R (April 2006). "Androgen physiology". Semin. Reprod. Med. 24 (2): 71–7.
• Ly LP, Jimenez M, Zhuang TN, Celermajer DS, Conway AJ, Handelsman DJ. A double-blind, placebo-controlled, randomized clinical trial of transdermal dihydrotestosterone gel on muscular strength, mobility, and quality of life in older men with partial androgen deficiency.
• Cailleux-Bounacer A, Rohmer V, Lahlou N, Lefebvre H, Roger M, Kuhn JM.
• Impact level of dihydrotestosterone on the hypothalamic-pituitary-leydig cell axis in men. Int J Androl. 2007 Oct 11.
• Kunelius P, Lukkarinen O, Hannuksela ML, Itkonen O, Tapanainen. The effects of transdermal dihydrotestosterone in the aging male: a prospective, randomized, double blind study. JSJ Clin Endocrinol Metab. 2002 Apr;87(4):1467-72.
• de Lignieres B. Transdermal dihydrotestosterone treatment of 'andropause'. Ann Med. 1993 Jun;25(3):235-41.
• Sakhri S, Gooren LJ. Safety aspects of androgen treatment with 5alpha-dihydrotestosterone. Andrologia. 2007 Dec;39(6):216-22.
• Boada LD, Fernández L, Zumbado M, Luzardo OP, Chirino R, Díaz-Chico BN.
• Identification of a specific binding site for the anabolic steroid stanozolol in male rat liver microsomes. J Pharmacol Exp Ther. 1996 Dec;279(3):1123-9.
• Fernández L, Chirino R, Boada LD, Navarro D, Cabrera N, del Rio I, Díaz-Chico BN. Stanozolol and danazol, unlike natural androgens, interact with the low affinity glucocorticoid-binding sites from male rat liver microsomes. Endocrinology. 1994 Mar;134(3):1401-8.
• Guillon G. Experiment with mesterolone in male fertility disorders Z Hautkr. 1975 Apr 1;50(7):293-7.
• Szöllösi J, Falkay GY, Sas M. Mesterolone treatment of patients with pathospermia. Int Urol Nephrol. 1978;10(3):251-6.
• Nikkanen V. Plasma cholesterol, triglycerides, FSH and testosterone levels of normolipemic male patients with decreased fertility treated with mesterolone. Andrologia. 1979 Jan;11(1):33-6.
• Jackaman FR, Ansell ID, Ghanadian R, McLoughlin PV, Lewis JG, Chisholm GD.
• The hormone response to a synthetic androgen (mesterolone) in oligospermia.
• Clin Endocrinol (Oxf). 1977 May;6(5):339-45.
• Christiansen, K. in Testosterone Action, Deficiency, and Substitution, Nieschlag, E. and Behre, H.M. eds. Springer-Verlag, New York, 107-142, 1998.
STUDIES/CLINICAL INFORMATION:
• Kuhnz W, Fritzemeier KH, Hegele-Hartung C, Krattenmacher R. Comparative progestational activity of norgestimate, levonorgestrel-oxime and levonorgestrel in the rat and binding of these compounds to the progesterone receptor. Contraception. 1995 Feb;51(2):131-9.
• Li QG, Li DZ. Analysis of anti-implantational action of norethisterone-3-oxime. Contraception. 1987 Dec;36(6):667-76.
• Li DZ, Fu GM, Zuo MD, Zhou MY, Zhao XL, Wang GP, Wei ZN. Pharmacokinetic studies of norethisterone-3-oxime and norethisterone-3-oxime acetate in rhesus monkey. J Tongji Med Univ. 1991;11(2):81-7.
• Li QG. Aromatization and hydrolysis of norethisterone-3-oxime in rabbit. J Steroid Biochem Mol Biol. 1994 Jun;49(2-3):227-31.
• Hammerstein J. Prodrugs: advantage or disadvantage? Am J Obstet Gynecol. 1990 Dec;163(6 Pt 2):2198-203.
• Shrivastav TG, Basu A, Kariya KP. One step enzyme linked immunosorbent assay for direct estimation of serum testosterone. J Immunoassay Immunochem. 2003;24(2):205-17.
• Tripathi V, Nara S, Chaube SK, Rangari K, Saroha A, Kariya KP, Singh H, Shrivastav TG. J Development of rapid and sensitive one-step direct enzyme linked immunosorbent assay for 17-alpha-OH-progesterone in serum. Immunoassay Immunochem. 2008; 29(2):117-27.
• Nara S, Tripathi V, Chaube SK, Rangari K, Singh H, Kariya KP, Shrivastav TG. A novel enzyme-linked immunosorbent assay for cortisol using a long-chain biotinylated cortisol-3-CMO derivative. J Immunoassay Immunochem. 2008;29(4):390-405.
• Sinnecker G, Köhler S Sex hormone-binding globulin response to the anabolic steroid stanozolol: evidence for its suitability as a biological androgen sensitivity test. J Clin Endocrinol Metab. 1989 Jun;68(6):1195-200.
• Saartok T, Dahlberg E, Gustafsson JA. Relative binding affinity of anabolic-androgenic steroids: comparison of the binding to the androgen receptors in skeletal muscle and in prostate, as well as to sex hormone-binding globulin. Endocrinology. 1984 Jun;114(6):2100-6.
• Tóth M, Zakár T. Relative binding affinities of testosterone, 19-nortestosterone and their 5 alpha-reduced derivatives to the androgen receptor and to other androgen-binding proteins: a suggested role of 5 alpha-reductive steroid metabolism in the dissociation of "myotropic" and "androgenic" activities of 19-nortestosterone. J Steroid Biochem. 1982 Dec;17(6):653-60.
• Lu NZ, Wardell SE, Burnstein KL, Defranco D, Fuller PJ, Giguere V, Hochberg RB, McKay L, Renoir JM, Weigel NL, Wilson EM, McDonnell DP, Cidlowski JA (December 2006). "International Union of Pharmacology. LXV. The pharmacology and classification of the nuclear receptor superfamily: glucocorticoid, mineralocorticoid, progesterone, and androgen receptors". Pharmacol. Rev. 58 (4): 782–97.
• Hammond GL, Abrams LS, Creasy GW, Natarajan J, Allen JG, Siiteri PK.
• Serum distribution of the major metabolites of norgestimate in relation to its
• pharmacological properties. Contraception. 2003 Feb;67(2):93-9.
• Juchem M, Pollow K, Elger W, Hoffmann G, Möbus V. Receptor binding of norgestimate--a new orally active synthetic progestational compound. Contraception. 1993 Mar;47(3):283-94.
• Ouyang G, Chen Z, Cai XJ, Song BA, Bhadury PS, Yang S, Jin LH, Xue W, Hu DY, Zeng S. Synthesis and antiviral activity of novel pyrazole derivatives containing oxime esters group. Bioorg Med Chem. 2008 Nov 15;16(22):9699-707. Epub 2008 Oct 2.
• Shrivastav TG, Kanaujia PK. Direct radioimmunoassay for the measurement of serum testosterone using 3H as label. J Immunoassay Immunochem. 2007;28(2):127-36.
• Basu A, Shrivastav TG One step enzyme linked immunosorbent assay for direct estimation of serum cortisol. J Immunoassay. 2000 Feb;21(1):39-50.
• Koji Sato, Motoyuki Iemitsu, Katsuji Aizawa, and Ryuichi Ajisaka Testosterone and DHEA activate the glucose metabolism-related signaling pathway in skeletal muscle. Am J Physiol Endocrinol Metab 294: E961-E968, 2008.
• Davison SL, Bell R (April 2006). "Androgen physiology". Semin. Reprod. Med. 24 (2): 71–7.
• Ly LP, Jimenez M, Zhuang TN, Celermajer DS, Conway AJ, Handelsman DJ. A double-blind, placebo-controlled, randomized clinical trial of transdermal dihydrotestosterone gel on muscular strength, mobility, and quality of life in older men with partial androgen deficiency.
• Cailleux-Bounacer A, Rohmer V, Lahlou N, Lefebvre H, Roger M, Kuhn JM.
• Impact level of dihydrotestosterone on the hypothalamic-pituitary-leydig cell axis in men. Int J Androl. 2007 Oct 11.
• Kunelius P, Lukkarinen O, Hannuksela ML, Itkonen O, Tapanainen. The effects of transdermal dihydrotestosterone in the aging male: a prospective, randomized, double blind study. JSJ Clin Endocrinol Metab. 2002 Apr;87(4):1467-72.
• de Lignieres B. Transdermal dihydrotestosterone treatment of 'andropause'. Ann Med. 1993 Jun;25(3):235-41.
• Sakhri S, Gooren LJ. Safety aspects of androgen treatment with 5alpha-dihydrotestosterone. Andrologia. 2007 Dec;39(6):216-22.
• Boada LD, Fernández L, Zumbado M, Luzardo OP, Chirino R, Díaz-Chico BN.
• Identification of a specific binding site for the anabolic steroid stanozolol in male rat liver microsomes. J Pharmacol Exp Ther. 1996 Dec;279(3):1123-9.
• Fernández L, Chirino R, Boada LD, Navarro D, Cabrera N, del Rio I, Díaz-Chico BN. Stanozolol and danazol, unlike natural androgens, interact with the low affinity glucocorticoid-binding sites from male rat liver microsomes. Endocrinology. 1994 Mar;134(3):1401-8.
• Guillon G. Experiment with mesterolone in male fertility disorders Z Hautkr. 1975 Apr 1;50(7):293-7.
• Szöllösi J, Falkay GY, Sas M. Mesterolone treatment of patients with pathospermia. Int Urol Nephrol. 1978;10(3):251-6.
• Nikkanen V. Plasma cholesterol, triglycerides, FSH and testosterone levels of normolipemic male patients with decreased fertility treated with mesterolone. Andrologia. 1979 Jan;11(1):33-6.
• Jackaman FR, Ansell ID, Ghanadian R, McLoughlin PV, Lewis JG, Chisholm GD.
• The hormone response to a synthetic androgen (mesterolone) in oligospermia.
• Clin Endocrinol (Oxf). 1977 May;6(5):339-45.
• Christiansen, K. in Testosterone Action, Deficiency, and Substitution, Nieschlag, E. and Behre, H.M. eds. Springer-Verlag, New York, 107-142, 1998.
Is The One going the be its final title?? lol
It has been mentioned to you before that your best chance is to order from AN direct, or from somone who WILL send to Australia (i.e. Nutra Planet).
I realize that, but in a PM it was also mentioned to me that AN would have to look at THEIR risk factor before sending, hence why I asked again.
I realize that, but in a PM it was also mentioned to me that AN would have to look at THEIR risk factor before sending, hence why I asked again.
